A blog that publishes updates and open access scientific papers about allergy, asthma and immunology. Editor: Juan Carlos Ivancevich, MD. Specialist in Allergy & Immunology
September 6, 2024
Aspirin Desensitization in Treatment of Chronic Rhinosinusitis with Nasal Polyposis: Current Protocols and Evidence
September 5, 2024
Recent Advances in Allergic Rhinitis: A Narrative Review.
Abstract
Allergic rhinitis (AR) is a prevalent chronic respiratory condition characterized by nasal inflammation, sneezing, congestion, and itching, significantly impacting quality of life. Over recent years, considerable advancements have been made in understanding the pathophysiology, diagnosis, and management of AR. This narrative review aims to synthesize these recent developments, providing a comprehensive overview of key areas. Emerging insights into AR pathophysiology have elucidated the complex interplay between genetic predisposition, environmental factors, and immune system dysregulation. Notably, the role of the epithelial barrier and the microbiome in AR pathogenesis has garnered increasing attention, offering potential targets for novel therapies. Advances in diagnostic technologies, such as component-resolved diagnostics and molecular allergology, have enhanced the precision of allergy identification, enabling more personalized treatment approaches.
Shared genetic architecture between gastro-esophageal reflux disease, asthma, and allergic diseases
Abstract
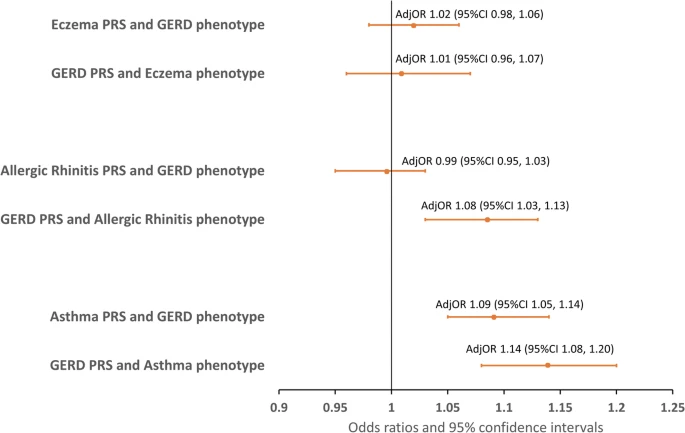 |
Associations between phenotypes and polygenic risk scores for GERD with eczema, allergic rhinitis, and asthma in Swedish twins. |
September 3, 2024
Real-World Study of Ragweed Sublingual Immunotherapy in Hungary
Abstract
Background: Ragweed (Ambrosia elatior) has become invasive in Europe, causing significant respiratory issues. Subcutaneous allergen immunotherapy (SCIT) has long been used to manage pollen allergies, but sublingual immunotherapy (SLIT) has gained interest.
Objective: This study aimed to evaluate the clinical benefits of ragweed SLIT under real-world in a cohort of Hungarian patients allergic to ragweed pollen.
Methods: We retrospectively reviewed the clinical records of 57 patients during the 2015 and 2016 ragweed pollen seasons. Patients were divided into two groups: Group 1 (n = 29), who had not received immunotherapy, and Group 2 (n = 28), who had previously undergone immunotherapy with another sublingual preparation. All patients were treated with Oraltek® ragweed for 4–6 months, initiating 2–4 months before the pollen season and rest of the period was 2 months of the 2016 pollen season. Symptom score (SS), medication score (MS), and combined symptom and medication score (CSMS) were evaluated intra- and intergroup.
Predictive models and applicability of artificial intelligence-based approaches in drug allergy
Núñez, Rafaela; Doña, Inmaculada; Cornejo-García, José Antonio. Current Opinion in Allergy and Clinical Immunology 24(4):p 189-194, August 2024. | DOI: 10.1097/ACI.0000000000001002
Abstract
Purpose of review
Drug allergy is responsible for a huge burden on public healthcare systems, representing in some instances a threat for patient's life. Diagnosis is complex due to the heterogeneity of clinical phenotypes and mechanisms involved, the limitations of in vitro tests, and the associated risk to in vivo tests. Predictive models, including those using recent advances in artificial intelligence, may circumvent these drawbacks, leading to an appropriate classification of patients and improving their management in clinical settings.
Recent findings
 |
| Graphical summary of the development and implementationof a ML model to diagnose drug allergy patients. |
September 2, 2024
Characterizing Long COVID in Children and Adolescents
Gross RS, Thaweethai T, Kleinman LC, et al. JAMA. Published online August 21, 2024. doi:10.1001/jama.2024.12747
Key Points
Question What prolonged symptoms experienced by youth are most associated with SARS-CoV-2 infection?Findings Among 5367 participants in the RECOVER-Pediatrics cohort study, 14 symptoms in both school-age children (6-11 years) and adolescents (12-17 years) were more common in those with vs without SARS-CoV-2 infection history, with 4 additional symptoms in school-age children only and 3 in adolescents only. Empirically derived indices for PASC research and associated clustering patterns were developed.
Meaning This study developed research indices for characterizing pediatric PASC. Symptom patterns were similar but distinguishable between school-age children and adolescents, highlighting the importance of characterizing PASC separately in different age groups.
Objective To identify the most common prolonged symptoms experienced by children (aged 6 to 17 years) after SARS-CoV-2 infection, how these symptoms differ by age (school-age [6-11 years] vs adolescents [12-17 years]), how they cluster into distinct phenotypes, and what symptoms in combination could be used as an empirically derived index to assist researchers to study the likely presence of PASC.
Design, Setting, and Participants Multicenter longitudinal observational cohort study with participants recruited from more than 60 US health care and community settings between March 2022 and December 2023, including school-age children and adolescents with and without SARS-CoV-2 infection history.
Exposure SARS-CoV-2 infection.
Main Outcomes and Measures PASC and 89 prolonged symptoms across 9 symptom domains.
 |
| Development of the Postacute Sequelae of SARS-CoV-2 Infection (PASC) Research Index and Threshold for Adolescents (Ages 12 to 17 years) |
Differential decline of SARS-CoV-2-specific antibody levels, innate and adaptive immune cells, and shift of Th1/inflammatory to Th2 serum cytokine levels long after first COVID-19
Abstract
Background
SARS-CoV-2 has triggered a pandemic and contributes to long-lasting morbidity. Several studies have investigated immediate cellular and humoral immune responses during acute infection. However, little is known about long-term effects of COVID-19 on the immune system.
Methods
We performed a longitudinal investigation of cellular and humoral immune parameters in 106 non-vaccinated subjects ten weeks (10 w) and ten months (10 m) after their first SARS-CoV-2 infection. Peripheral blood immune cells were analyzed by multiparametric flow cytometry, serum cytokines were examined by multiplex technology. Antibodies specific for the Spike protein (S), the receptor-binding domain (RBD) and the nucleocapsid protein (NC) were determined. All parameters measured 10 w and 10 m after infection were compared with those of a matched, noninfected control group (n = 98).
Results
 |
| Study enrolment scheme and antibody decline pattern. |
August 31, 2024
Allergen immunotherapy adverse events in adults with respiratory allergies-data from ADER: An EAACI task force report
Abstract
Background
Registries can yield important insights on allergen immunotherapy (AIT) outcomes in daily clinical practice. However, systematic recordings of adverse events (AE) due to AIT in real-life are lacking.
Methods
The Allergen Immunotherapy Adverse Events Registry (ADER) is a prospective, multicenter registry on real-life AIT safety. Data on adults (>18 years old) with respiratory allergies receiving AIT with mites, pollens, epithelia, and/or molds were retrieved and analyzed from ADER. The frequency, characteristics and risk factors of AE were investigated. The MedDRA terminology was used to record AE.Results
A total of 1545 individuals with a mean age of 33 ± 10 years receiving 1815 AIT courses (n = 1060 sublingual (SLIT); n = 755 subcutaneous (SCIT)) in centers from eight countries were included. Patients had allergic rhinitis (65%) or, asthma only (3.7%) or rhinitis with asthma (31.2%).






