Abstract
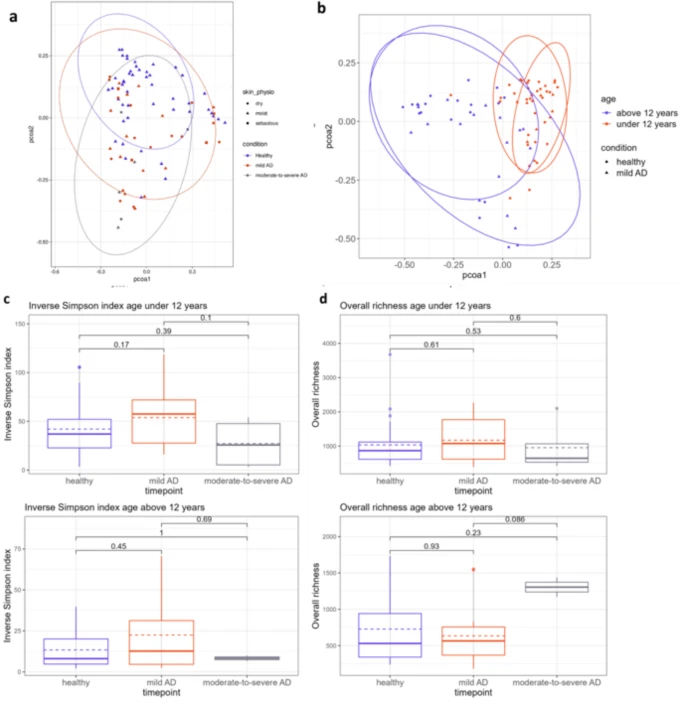 |
Associations between atopic dermatitis and skin microbiome diversity |
A blog that publishes updates and open access scientific papers about allergy, asthma and immunology. Editor: Juan Carlos Ivancevich, MD. Specialist in Allergy & Immunology
Abstract
 |
Associations between atopic dermatitis and skin microbiome diversity |
Abstract
Managing pruritic conditions is essential due to their significant impact on patients' quality of life. Chronic urticaria (CU), characterized by persistent itching and hives, severely affects daily activities and sleep. CU includes chronic inducible urticaria and chronic spontaneous urticaria, with the latter lacking identifiable triggers, making treatment especially challenging. CU is a condition that occurs across all age groups, with a higher prevalence among young adults and middle-aged women.
 |
| Clinical profile differences between various second-generation H1 antihistamines |
Buchheit
Abstract
 |
| IL-5 is a key cytokine for type 2 inflammation and plays a broad role in pathophysiology beyond eosinophilic inflammation. |
Abstract
 |
| The comparison of medications used by participants versus medications recommended by guidelines appropriate for the participants’ AR status (n = 167). |
Batard, T., Taillé, C., Guilleminault, L., Bozek, A., Floch, V.-L., Pfaar, O., Canonica, W., Akdis, C., Shamji, M. and Mascarell, L. (2024) Clin Exp Allergy. https://doi.org/10.1111/cea.14575
ABSTRACT
 |
| Graphical Abstract |
Sladek S, Unger-Manhart N, Siegl C et al. Clin Ophthalmol. 2024;18:2797-2811 https://doi.org/10.2147/OPTH.S476163
Abstract:
Purpose: This randomized, placebo-controlled, crossover, double‐blind trial aimed to evaluate the efficacy and safety of Tacrosolv, a novel 0.005% tacrolimus eye-drop solution, in adults with grass pollen–induced allergic conjunctivitis.
Methods: A total of 64 adult participants were randomized to receive 2.5 μg or 5 μg tacrolimus/eye/day or placebo treatment for 8 days, with grass pollen exposure on day 1 and day 8. After a 2-week washout period, placebo participants crossed over to Tacrosolv treatment and vice versa, with repeated treatment and exposure. During exposure, participants recorded ocular, nasal, and respiratory allergy symptoms every 15 minutes. The primary endpoint was the mean total ocular symptom score (TOSS) on day 8. Objective ocular safety parameters were assessed before, during, and after exposure. Adverse events were recorded throughout the study.