Summary
 |
| Biomarkers and anti-inflammatory strategies in type 2 diseases. |
A blog that publishes updates and open access scientific papers about allergy, asthma and immunology. Editor: Juan Carlos Ivancevich, MD. Specialist in Allergy & Immunology
Summary
 |
| Biomarkers and anti-inflammatory strategies in type 2 diseases. |
Hossenbaccus, L., Walker, T. & Ellis, A.K. Allergy Asthma Clin Immunol 21, 6 (2025). https://doi.org/10.1186/s13223-024-00928-1
To the Editor,
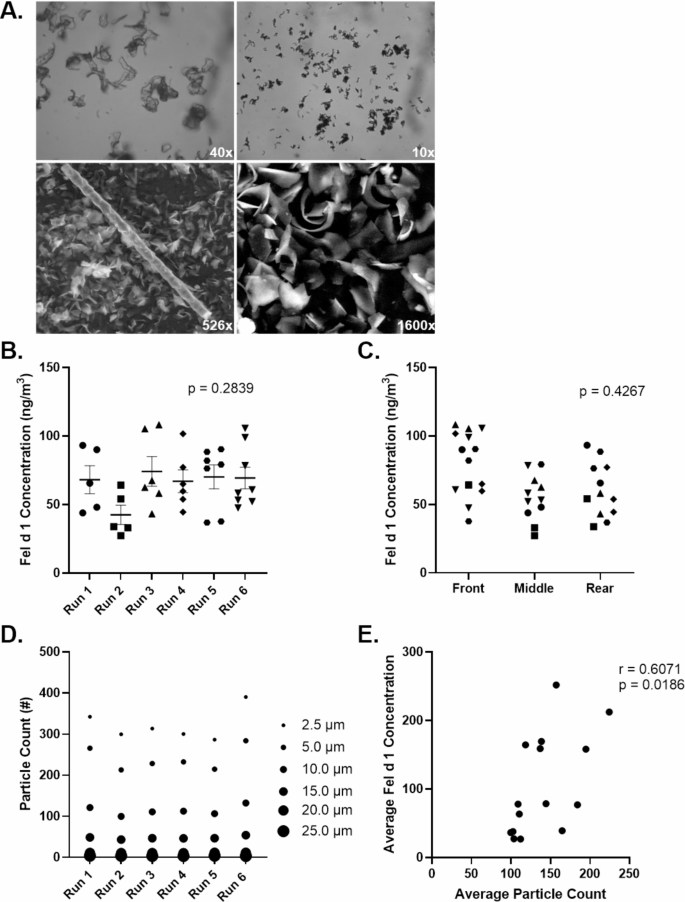 |
| Consistent Fel d 1 concentrations and particle counts were observed across runs in the Specialized Particulate ControlEnvironmental Exposure Unit. |
For the study of perennial allergens, the Specialized Particulate Control Environmental Exposure Unit (SPaC-EEU; formerly known as the house dust mite (HDM)-EEU) has been established.
Frau, F., Loustalot, P., Törnqvist, M. et al. Sci Rep 15, 3082 (2025). https://doi.org/10.1038/s41598-024-78794-5
Abstract
 |
| UMAP projections in two dimensions (a) and three dimensions (b, c, d)of all clusters of AD patients |
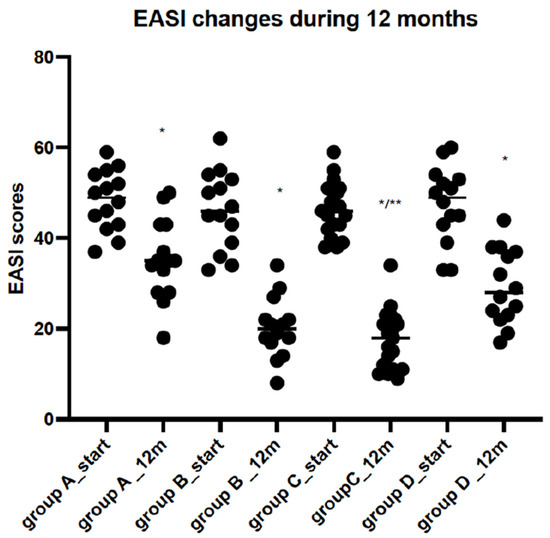 |
| EASI score changes before and after 12 months of observation. |
Abstract
Background
Chronic rhinosinusitis with nasal polyps (CRSwNP) is a recurrent inflammatory disease associated with several comorbidities and a significant disease burden for patients. Treatments include corticosteroids and sinonasal surgery, but these can be associated with the risk of adverse events and nasal polyp recurrence. Biologic treatments such as mepolizumab can be used as an add-on treatment and are effective at reducing surgery and corticosteroid use.
Main text
 |
| Patient scenarios illustrating where the MDT approach may be applied for patients with CRSwNP and comorbid severe asthma |
Jeimy, S., Wong, T., Ben-Shoshan, M. et al. Allergy Asthma Clin Immunol 20 (Suppl 3), 78 (2024). https://doi.org/10.1186/s13223-024-00936-1
Abstract
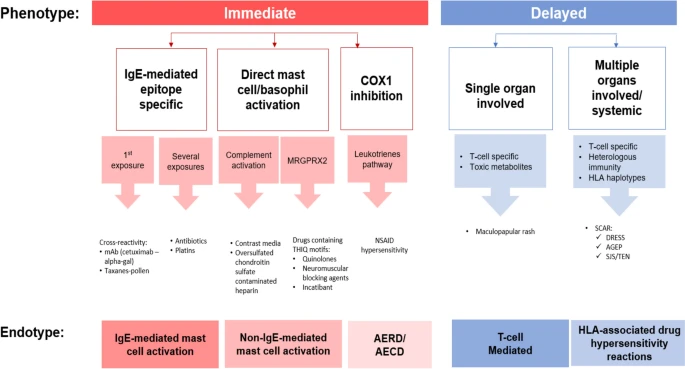 |
| Drug allergy classification based on phenotypes and endotypes |
Background
Until recently, immediate emergency department (ED) transfer after food-related anaphylactic reactions was recommended regardless of symptom resolution following use of an epinephrine autoinjector (EAI). We evaluated the cost-effectiveness of delayed ED transfer after EAI use in non-medical settings (watchful waiting) compared to immediate ED transfer among pediatric patients with food allergies in Canada.
Methods
We developed a probabilistic Markov model of individuals starting at age of one year who are at risk of severe food-related allergic reactions requiring epinephrine. We evaluated medical costs (in 2022 Canadian dollars) and quality-adjusted life years (QALY) of each strategy over a 20-year horizon. In the base case, we assumed a tenfold increase in food allergy fatality for patients under watchful waiting, which we increased to 100- to 1,000-fold in sensitivity analysis.