Abstract
Anaphylaxis is a serious hypersensitivity reaction that may be life-threatening. Data suggest that fatal anaphylaxis is a very rare event; however, anaphylaxis and fear of anaphylaxis are associated with high societal burden and cost. Cost-effectiveness analyses can facilitate a model of value-based care; in this review, we aim to highlight key health-economic analyses in anaphylaxis. Health economic evaluations suggest that screening approaches to prevent anaphylaxis have low value.A blog that publishes updates and open access scientific papers about allergy, asthma and immunology. Editor: Juan Carlos Ivancevich, MD. Specialist in Allergy & Immunology
September 22, 2025
Cost-effective care in anaphylaxis prevention and management
Investigating the Use of Melatonin in Children With Atopic Dermatitis: A Cross-Sectional Study
ABSTRACT
Background/Objectives
Children with atopic dermatitis (AD) frequently experience sleep disturbances. Melatonin, an over-the-counter supplement, is increasingly used in the general pediatric population; however, its prevalence and perceived effectiveness in children with AD remain unclear.
Methods
Caregivers of pediatric patients (ages 1–17 years) with physician-diagnosed AD completed surveys on melatonin use, Patient Oriented Eczema Measure (POEM), and age-appropriate sleep questionnaires.
Results
Among 102 total respondents, 27.5% reported a history of melatonin use, with 10.8% using it in the past 30 days. Gummies were the most common formulation (85.7%), though brands and dosages varied widely (0.5–10 mg; median: 3 mg).Comparative Efficacy of Montelukast-Levocetirizine Combination Therapy versus Monotherapy in Allergic Rhinitis: A Systematic Review and Meta-Analysis
Abstract
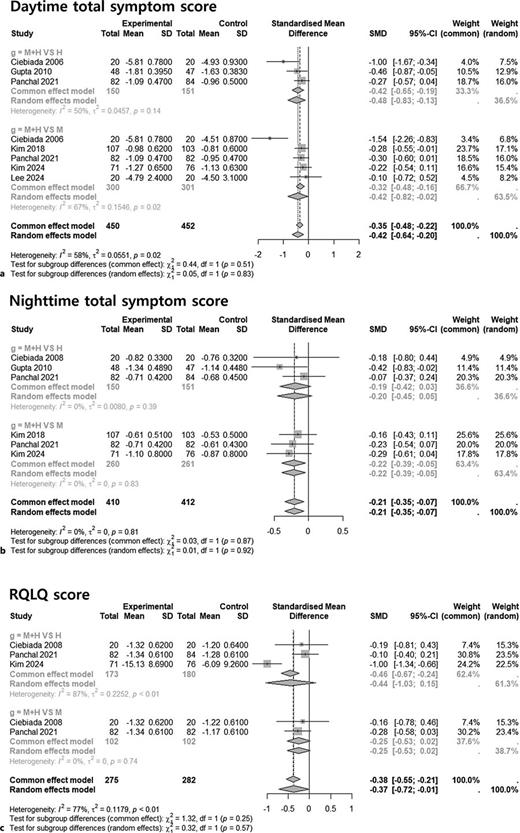
Introduction: Combination therapy with antihistamines and leukotriene receptor antagonists (LTRAs) is frequently used in allergic rhinitis (AR), particularly in patients with persistent or inadequately controlled symptoms. However, supporting evidence has been inconsistent. This meta-analysis assessed the efficacy of montelukast combined with levocetirizine in patients with AR by directly comparing the combination therapy with each monotherapy and evaluating individual symptom domains.
Methods: A systematic review and meta-analysis of randomized controlled trials (RCTs) was conducted, with database searches performed through March 2025. Outcomes included daytime and nighttime total symptom scores, individual nasal symptoms, rhinoconjunctivitis-related quality of life (RQLQ), and treatment-emergent adverse events (TEAEs). Pooled effects were calculated using standardized mean differences (SMDs) and odds ratios (ORs) with 95% confidence intervals (CIs).
Results: Seven RCTs (n = 887) were included. Combination therapy resulted in greater improvement in daytime symptoms (SMD [95% CI]: −0.42 [–0.64 to −0.20]), rhinorrhea (SMD [95% CI]: −0.69 [–1.09 to −0.30]), and nasal congestion (SMD [95% CI]: −0.64 [–1.05 to −0.24]) compared to monotherapy.
September 20, 2025
Expert Perspectives on Next Generation Health Guidelines: How to Integrate RWE in EBM
Abstract:
Integrating real-world evidence (RWE) into evidence-based medicine (EBM) enhances healthcare decision-making. RWE provides insights into the real-world effectiveness and safety of therapies and health technologies, filling gaps that clinical trials may leave. EBM, which concentrates on therapeutic issues, depends on rigorous evaluation of evidence, including data from randomized controlled trials (RCTs) and RWE. Combining evidence from RCTs and RWE when forming recommendations offers a comprehensive understanding of benefits and risks by considering their strengths, limitations, and standardized methods. The 2nd European Academy of Allergy & Clinical Immunology/Respiratory Effectiveness Group (EAACI/REG) Workshop, held in Rome, Italy, on October 4th, 2023, discussed integrating RWE and EBM. The goals were to develop recommendations for high-quality RWE and its inclusion in evidence syntheses, with a particular focus on airway diseases.
September 19, 2025
Subcutaneous Allergen Immunotherapy in Adults Allergic to House Dust Mites: A Phase 3 Randomized Controlled Trial
C. Bachert, H. Hoogeveen, R. ten Have, et al. Allergy (2025): 1–10, https://doi.org/10.1111/all.70063.
ABSTRACT
Background
Subcutaneous allergen immunotherapy (SCIT) has a longstanding history as a safe and effective treatment. Nevertheless, to meet the European Medicines Agency's regulatory requirements for continued market authorization, its efficacy and safety must be confirmed in a pivotal Phase III trial.
 |
| Graphical Abstract |
Based on a successful Phase II dose-finding study, the aim was to confirm the safety and efficacy of a subcutaneous house dust mites (HDM) preparation in a randomized controlled trial using an optimal higher dose than the current maintenance dose.
Method
Seven hundred sixty-seven subjects were randomized in a 1:1 ratio to 1-year treatment with SCIT-product at a dose of 50,000 AUeq/mL or placebo.
September 17, 2025
Proceedings of the Second Artificial Intelligence in Primary Immunodeficiencies (AIPI) Meeting
 |
| AI Integration Pathways in Primary Immunodeficiencies. Radial diagram summarizing the meeting discussion themes |
Stapokibart for Severe Uncontrolled Chronic Rhinosinusitis With Nasal Polyps The CROWNS-2 Randomized Clinical Trial
Shen S, Yan B, Wang M, et al. JAMA. 2025;334(11):962–972. doi:10.1001/jama.2025.12515
Key Points
Question Among patients with severe chronic rhinosinusitis with nasal polyps taking a daily intranasal corticosteroid, does stapokibart, a novel anti–interleukin 4Rα monoclonal antibody, decrease nasal polyps and reduce symptoms of nasal congestion?
Findings In this randomized clinical trial that included 179 participants from China, stapokibart significantly reduced polyp size and improved nasal symptoms at 24 weeks in patients with severe chronic rhinosinusitis with nasal polyps.
Meaning Among patients with severe chronic rhinosinusitis with nasal polyps treated with a daily intranasal corticosteroid, stapokibart reduced polyp size and severity of nasal symptoms at 24 weeks.
September 16, 2025
Validation of the Urticaria Activity Score for Cold Urticaria
Background
Cold urticaria (ColdU) is characterized by the appearance of wheals and/or angioedema and itch after exposure to cold stimuli. The Cold Urticaria Activity score (ColdUAS) is a newly devised patient-reported outcome measure (PROM) assessing disease activity in ColdU.
Objectives
We aimed to validate the ColdUAS according to PROM guidelines, assess the optimal documentation period, and develop a scoring computation algorithm.
Methods
We instructed 71 patients with typical and atypical ColdU to complete the ColdUAS questionnaire over 4 consecutive weeks and asked to fill out additional anchor instruments including global assessment tools and validated quality of life measures.