Kim JS, Stybayeva G, Hwang SH. Int Arch Allergy Immunol. 2025 Jul 15:1-14. doi: 10.1159/000547410.Abstract
Introduction: Combination therapy with antihistamines and leukotriene receptor antagonists (LTRAs) is frequently used in allergic rhinitis (AR), particularly in patients with persistent or inadequately controlled symptoms. However, supporting evidence has been inconsistent. This meta-analysis assessed the efficacy of montelukast combined with levocetirizine in patients with AR by directly comparing the combination therapy with each monotherapy and evaluating individual symptom domains.
Methods: A systematic review and meta-analysis of randomized controlled trials (RCTs) was conducted, with database searches performed through March 2025. Outcomes included daytime and nighttime total symptom scores, individual nasal symptoms, rhinoconjunctivitis-related quality of life (RQLQ), and treatment-emergent adverse events (TEAEs). Pooled effects were calculated using standardized mean differences (SMDs) and odds ratios (ORs) with 95% confidence intervals (CIs).
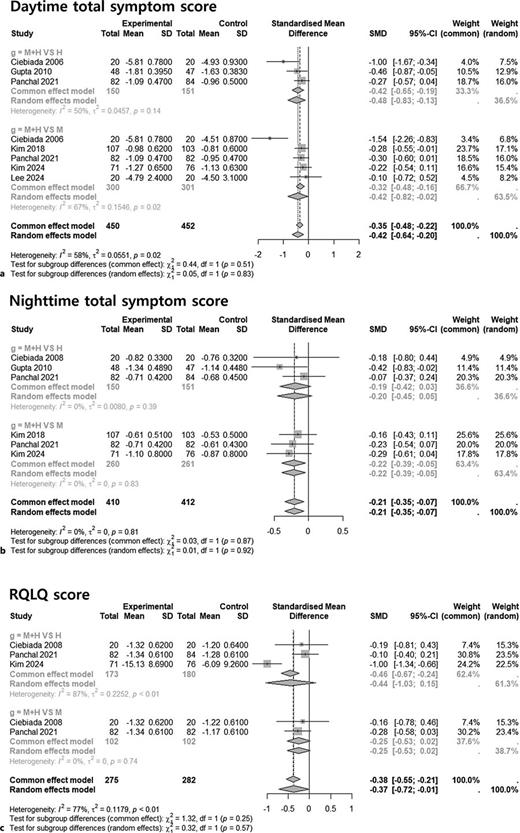 |
Forest plots comparing changes in daytime total symptom score (a),
nighttime total symptom score (b), and rhinoconjunctivitis quality of
lifequestionnaire (RQLQ) scores (c) between combination therapy
(montelukast-levocetirizine) and monotherapy. |
Results: Seven RCTs (n = 887) were included. Combination therapy resulted in greater improvement in daytime symptoms (SMD [95% CI]: −0.42 [–0.64 to −0.20]), rhinorrhea (SMD [95% CI]: −0.69 [–1.09 to −0.30]), and nasal congestion (SMD [95% CI]: −0.64 [–1.05 to −0.24]) compared to monotherapy.












