 |
| Approved biologic therapies for adults in Europe |
A blog that publishes updates and open access scientific papers about allergy, asthma and immunology. Editor: Juan Carlos Ivancevich, MD. Specialist in Allergy & Immunology
September 27, 2025
Biologics in the treatment of atopic dermatitis: approved active substances and monoclonal antibodies in advanced clinical trials
September 26, 2025
Artificial intelligence in clinical allergy practice: current status, challenges, and future directions
https://doi.org/10.37349/eaa.2025.100992
 |
| Summary of the applications of AI in clinical allergy practice |
Characterizing drug allergy management among allergists in Canada: a national survey study
Lee, E., Lee, B., Krishnan, S. et al. Allergy Asthma Clin Immunol 21, 41 (2025). https://doi.org/10.1186/s13223-025-00981-4
Abstract
Background
Unverified drug allergy labels are common and associated with significant patient harm, yet infrastructure and testing practices vary across clinical settings in Canada.
Objective
To characterize variability in drug allergy management among allergists in Canada and identify setting-specific barriers to drug allergy testing and desensitization.
Methods
We developed a peer-reviewed 40-item survey, distributed via the Canadian Society of Allergy and Clinical Immunology, to assess practice patterns, testing modalities, and perceived barriers among allergists. Descriptive statistics and Fisher’s exact test were used to evaluate responses by practice setting.
Results
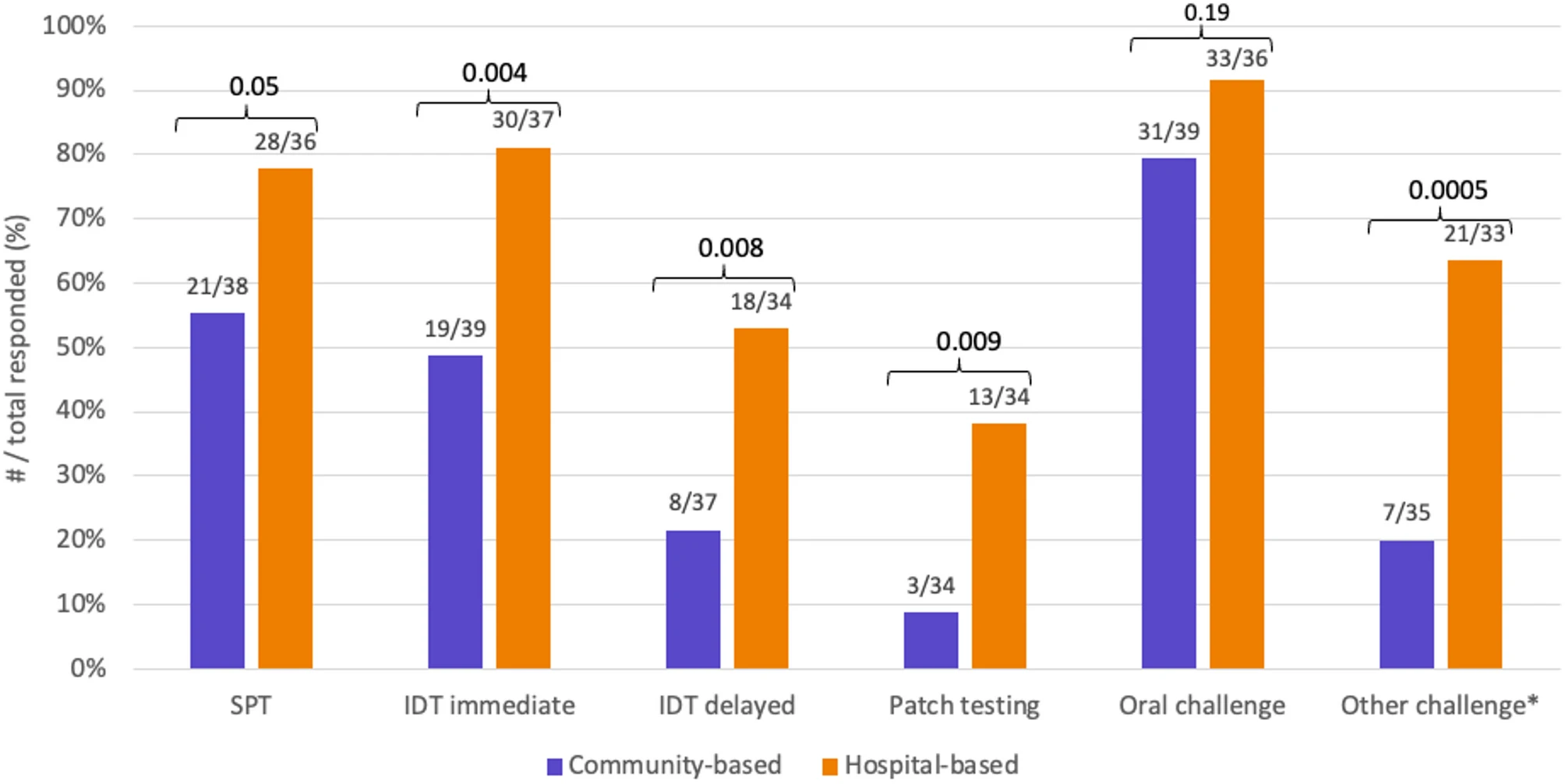 |
| Comparison of drug allergy testing modalities by practice setting |
September 25, 2025
What has changed with subcutaneous immunotherapy against house dust mites? Eight-year, single-center real-world data
Introduction:
We aimed to investigate clinical and laboratory parameters that can predict clinical response in patients who completed subcutaneous immunotherapy (SCIT) against house dust mites (HDM) and to determine parameters associated with systemic adverse effects (SAE).
Methods:
In total, 108 patients who had complete medical data were evaluated.
Results:
 |
| Laboratory values and symptom scores before and after SCIT |
Machine learning in allergy research: A bibliometric review
 |
| Word clouds for topics obtained by Latent Dirichlet allocation (LDA) |
September 24, 2025
The evolution of food protein-induced enterocolitis syndrome (FPIES): Global trends, emerging triggers, and natural history.
Anvari S, Gupta M, Nicolaides R et al. Ann Allergy Asthma Immunol. 2025 Sep 19:S1081-1206(25)01160-3. doi: 10.1016/j.anai.2025.09.006.
 |
| Graphical timeline of our understanding of FPIES. |
Social Determinants of Health in Pediatric Asthma and Allergic Diseases: A Systematic Review
Koumpagioti, D., Moriki, D., Boutopoulou, B.et al. (2025). Epidemiologia, 6(3), 56. https://doi.org/10.3390/epidemiologia6030056
Abstract
 |
| PRISMA diagram for the study selection process. |
September 23, 2025
Artificial Intelligence Diagnostic Accuracy and Clinical Utility in Allergic Rhinitis: Systematic Review
Dana A. Alrezq, Abdulaziz M. Aldaghmani, Jana J. Alali, et al. Authorea. September 12, 2025.
DOI: 10.22541/au.175766646.65353803/v1
Abstract
Allergic rhinitis (AR) is one of the most common outpatient conditions, diagnosed through operator-dependent and resource-intensive methodologies. Consequently, there has been a surge in the development of diagnostic strategies using artificial intelligence (AI). This review aims to explore how AI can enhance diagnostic accuracy, personalize treatment, and support clinical decisions in AR care. The review followed PRISMA guidelines and was registered in PROSPERO. PubMed, Cochrane Central, Embase, and Google Scholar were searched for studies published between 2002 and 2025. Out of 1,109 identified studies, eight studies met the inclusion criteria.





