Harvey-Seutcheu, C.; Hopkins, G.; Fairclough, L.C. Int. J. Mol. Sci. 2024, 25, 3255. https://doi.org/10.3390/ijms25063255
Abstract
 |
| Summary of cells of origin, characteristics, and effects of S. aureus-, M. sympodialis-, and mast cell-derived EVs. |
A blog that publishes updates and open access scientific papers about allergy, asthma and immunology. Editor: Juan Carlos Ivancevich, MD. Specialist in Allergy & Immunology
Harvey-Seutcheu, C.; Hopkins, G.; Fairclough, L.C. Int. J. Mol. Sci. 2024, 25, 3255. https://doi.org/10.3390/ijms25063255
Abstract
 |
| Summary of cells of origin, characteristics, and effects of S. aureus-, M. sympodialis-, and mast cell-derived EVs. |
Mir, A., Lanoue, D., Zanichelli, V. et al. Allergy Asthma Clin Immunol 20, 20 (2024). https://doi.org/10.1186/s13223-024-00877-9
Abstract
Background
Self-reported penicillin allergy labels are common and often inaccurate after assessment. These labels can lead to reduced use of first-line beta-lactam antibiotics and worse outcomes. We measured the impact of a previously performed inpatient proactive systematic penicillin allergy de-labelling program on subsequent antibiotic use. This prior program included assessment, risk-stratification, and low risk direct oral amoxicillin challenge.
Methods
 |
| Schematic representation of the de-labelling implementation program |
Results
A total of 368 patients with penicillin allergy label were included across two campuses and study periods. 24 (13.8%) patients in the intervention groups had sustained penicillin allergy label removal at 30 days from admission vs. 3 (1.5%) in the non-intervention group (p < 0.001).
Takala, J., Vähätalo, I., Tuomisto, L.E. et al. npj Prim. Care Respir. Med. 34, 2 (2024). https://doi.org/10.1038/s41533-024-00360-3
Abstract
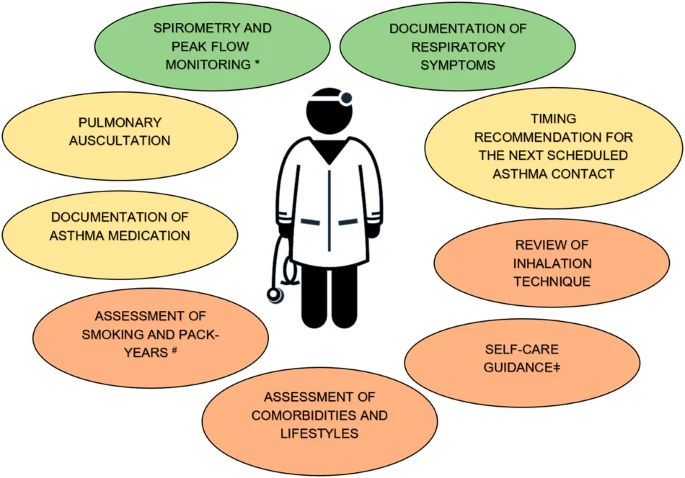 |
| Green colour describes the performed assessment that were implemented well, yellow colour describes moderate implementation, and the orange describes the measures that are poorly implemented. |
Lin CL, Lee KH, Huang WT, Hsieh LC, Wang CM. J Microbiol Immunol Infect. 2024 Feb;57(1):175-183. doi: 10.1016/j.jmii.2023.11.005.
Abstract
Background
Children with allergic rhinitis (AR) have substantially more acute rhinosinusitis than children without AR. We evaluated whether intranasal corticosteroids (INCS), second-generation antihistamines (SGH), and/or intranasal antihistamines (INH) for AR affect acute rhinosinusitis in children with AR aged 2–18 years.
Methods
 |
| Flow chart of participant enrollment. |
Abstract
Atopic dermatitis (AD) is a pervasive and multifaceted dermatological disorder causing daily distress to afflicted individuals worldwide. This comprehensive review synthesizes the historical and contemporary advancements in therapeutic strategies, offering a critical analysis of their efficacy, safety profiles, and adaptability. The enduring role of topical corticosteroids in managing AD is examined, acknowledging their potent anti-inflammatory properties alongside their potential adverse side effects, particularly in extended usage. The article explores the utilization of topical calcineurin inhibitors like tacrolimus and pimecrolimus, highlighting their novel anti-inflammatory pathways while also scrutinizing concerns over potential malignancies that relegate them to second-line therapy.
 |
| Comparative studies of PDE4 inhibitors, calcineurin inhibitors, and topical corticosteroids in the pharmacological management of atopic dermatitis |