Kapadia S, Nageswaramma S, Shah K, et al. Cureus 16(1): e53125. doi:10.7759/cureus.53125
Abstract
Background: Chronic spontaneous urticaria (CSU) is a debilitating affliction that affects diverse quality of life (QoL) parameters such as sleep, self-esteem, and daily activities. Second-generation antihistamines, such as desloratadine, are more effective and safer in managing CSU. Desloratadine is a nonsedating, potent, and highly selective H1 receptor antagonist. At its daily dose of 5 mg, almost half of CSU patients do not show symptomatic improvement. European Academy of Allergy and Clinical Immunology (EAACI)/Global Allergy and Asthma European Network (GA2LEN)/European Dermatology Forum (EDF) (EuroGuiDerm)/Asia Pacific Association of Allergy, Asthma and Clinical Immunology (APAAACI) guidelines recommend increasing the dosage to up to four times in such nonresponsive patients. However, there is insufficient clinical evidence in Indian settings.
Method: We evaluated the efficacy and safety of 10 mg desloratadine (OD) in 256 nonresponsive patients with moderate to severe CSU.
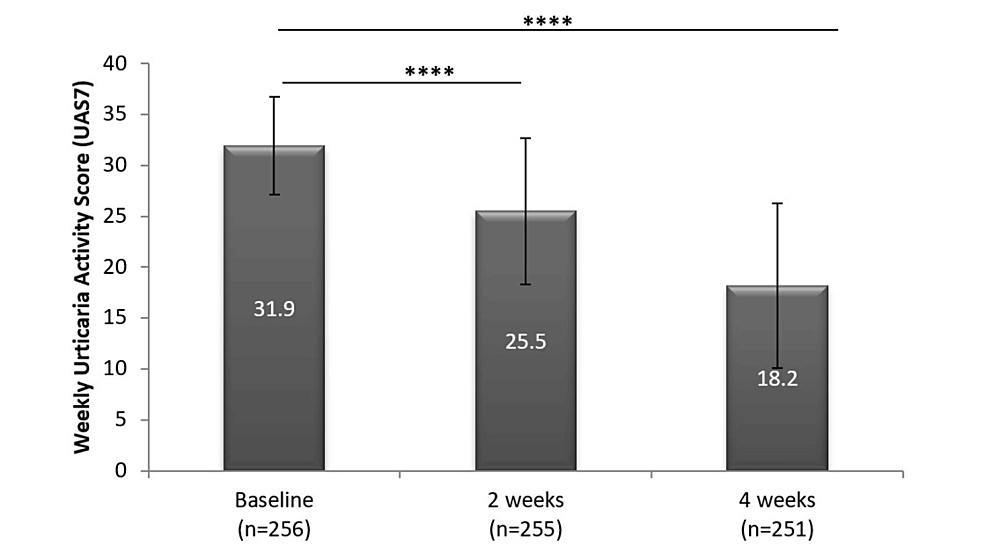
The primary outcome was the change in Urticaria Activity Score (UAS7) from baseline to four weeks. Additionally, change in Chronic Urticaria Quality of Life (CU-Q2oL) scores during the course of treatment was also evaluated.Result: The mean UAS7 scores showed a significant reduction from 31.9 ± 4.8 at baseline to 18.2 ± 8.1 at the end of the study (p < 0.0001). The use of a higher dose of desloratadine also decreased the CU-Q2oL scores significantly from 59.8 ± 14.7 at baseline to 35.4 ± 10 at four weeks (p < 0.0001). The incidence of adverse events (AEs) possibly linked to the drug was low (1.6%), and no serious adverse events were reported.Conclusion: Results indicated improvements in the disease severity as well as its positive impact on participants’ QoL. This study confirms the efficacy and safety of daily use of a twofold dose of desloratadine in nonresponsive moderate to severe CSU patients.

No comments:
Post a Comment