Research - Open access
Thomas B Casale, Thomas Corbridge, Guillaume Germain, François Laliberté, Sean D MacKnight, Julien Boudreau, Mei S Duh & Arijita Deb. Allergy, Asthma & Clinical Immunology volume 20, Article number: 25 (2024)
Abstract
Background
Systemic corticosteroid (SCS) use remains widespread among patients with severe asthma, despite associated complications.
Objective
Evaluate the association between cumulative SCS exposure and SCS-related complications in severe asthma.
Methods
This retrospective, longitudinal study used claims data from the Optum Clinformatics Data Mart database (GSK ID: 214469). Eligible patients (≥ 12 years old) had an asthma diagnosis and were divided into two cohorts: SCS use and non/burst-SCS use. Patients in the SCS use cohort had a claim for a daily prednisone-equivalent dose ≥ 5 mg SCS following ≥ 6 months of continuous SCS use; those in the non/burst-SCS cohort had no evidence of continuous SCS use and had a non-SCS controller/rescue medication initiation claim. For each cohort, the date of the qualifying claim was the index date. SCS users were further stratified by SCS use during each quarter of follow-up: low (≤ 6 mg/day), medium (> 6–12 mg/day), high (> 12 mg/day), and continuous high (≥ 20 mg/day for 90 days).
SCS-related complications were evaluated in the quarter following SCS exposure. The adjusted odds ratios (OR) of experiencing SCS-related complications during follow-up in each of the SCS use groups versus the non/burst SCS cohort were calculated using generalized estimating equations models.Results
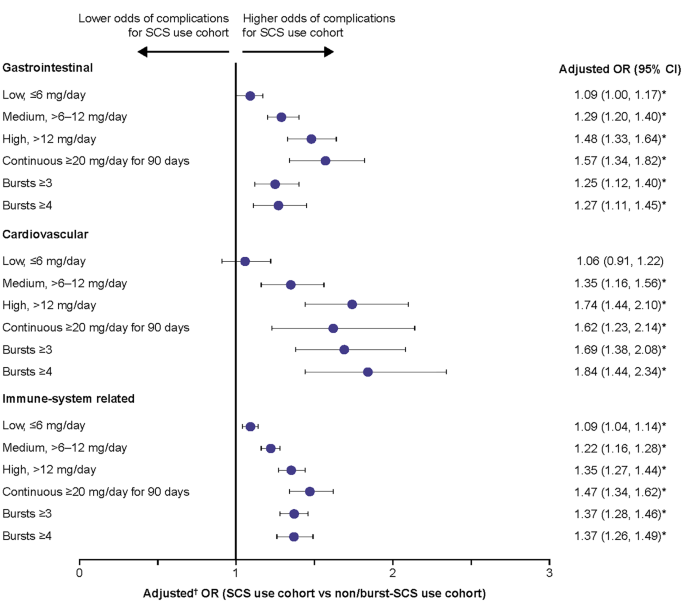 |
| Odds of acute SCS-related complications during the follow-up period due to SCS exposure. |
Conclusion
SCS use, even at low doses, is associated with increased risk of SCS-related complications among patients with severe asthma.

No comments:
Post a Comment