Catamerò F, Bragato MC, Lozano MA et al. Vaccines. 2026; 14(4):326. https://doi.org/10.3390/vaccines14040326
Abstract
Background/Objectives: Allergen immunotherapy (AIT), involving subcutaneous (SCIT) or sublingual (SLIT) administration of the culprit allergen, is the only treatment capable of modifying the natural course of allergic diseases, and provides lasting benefits in terms of symptom reduction and medication use. AIT for allergic rhinitis is acknowledged as safe and effective in both adults and children; however, no studies have comprehensively evaluated the safety and efficacy of AIT in these populations, integrating results from randomized controlled trials (RCTs) and real-world evidence (RWE).Methods: We evaluated data in the literature including studies from RCTs and RWE in which the safety and efficacy of AIT in both children and adults have been analyzed. A narrative literature search was conducted in PubMed up to January 2026 using the following keywords for the search string: “allergen immunotherapy,” “AIT,” “safety,” “efficacy,” “clinical outcome,” and “clinical evaluation.”
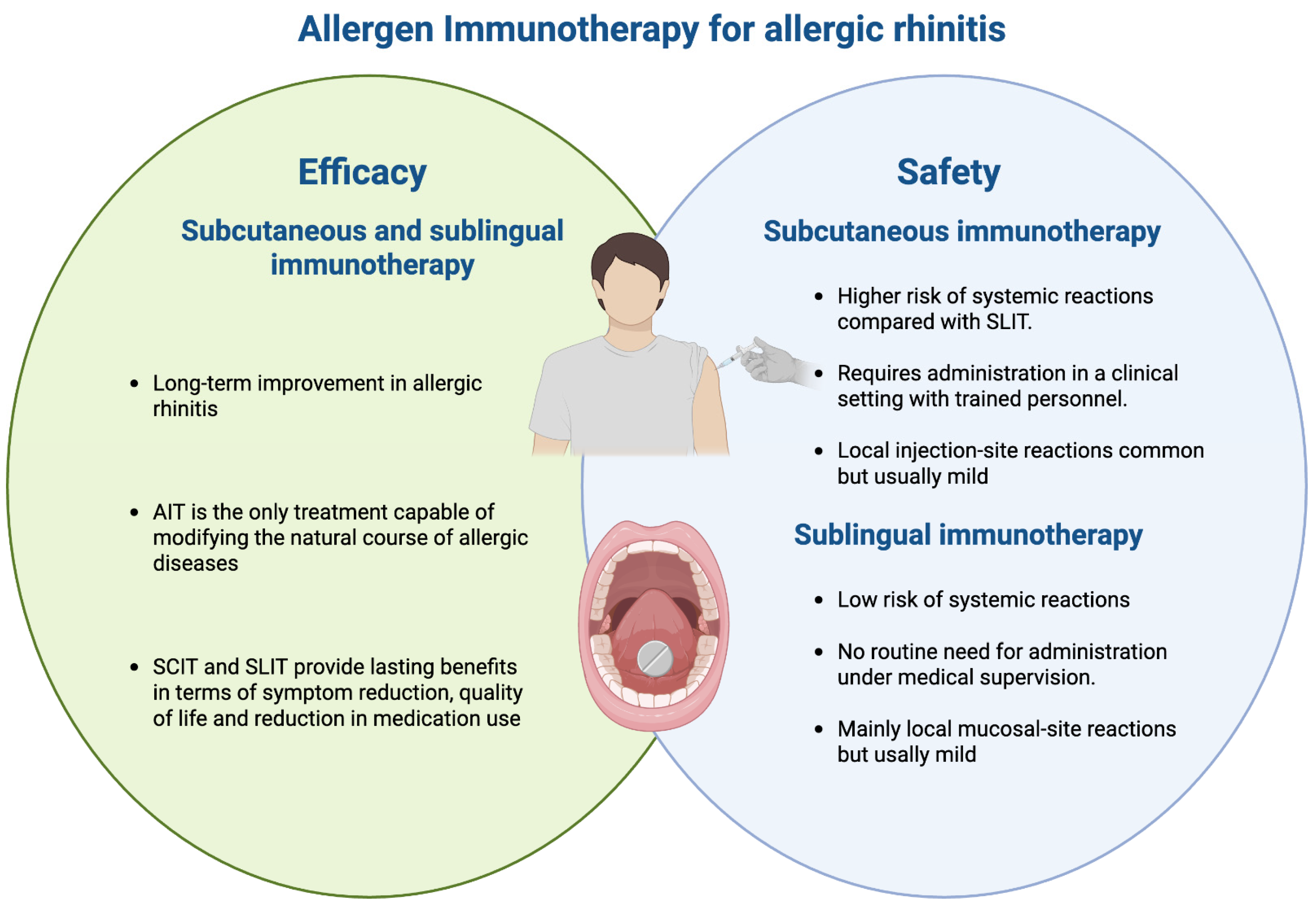 |
| Efficacy and safety of allergen immunotherapy in allergic rhinitis. AIT, allergen immunotherapy; SCIT, subcutaneous immunotherapy; SLIT, sublingual immunotherapy. |
Conclusions: AIT was effective and safe for treating allergic rhinitis across RCT and RWE studies. Integrating RWE with RCT findings is essential for guideline development, particularly for capturing long-term outcomes and real-world applications.
No comments:
Post a Comment