Abstract
 |
| Process of the 40-year-old male patient diagnose as TEN. |
Objectives: This study aimed to report the clinical outcomes of different types of SCARs treated with TNF-α inhibitors.
A blog that publishes updates and open access scientific papers about allergy, asthma and immunology. Editor: Juan Carlos Ivancevich, MD. Specialist in Allergy & Immunology
Abstract
 |
| Process of the 40-year-old male patient diagnose as TEN. |
Objectives: This study aimed to report the clinical outcomes of different types of SCARs treated with TNF-α inhibitors.
Santos AF, Riggioni C, Agache I, et al. Allergy. 2024; 00: 1-23. doi:10.1111/all.16345
Abstract
 |
| Management of IgE-mediated food allergy. |
This European Academy of Allergy and Clinical Immunology (EAACI) guideline provides recommendations for the management of IgE-mediated food allergy and was developed using the Grading of Recommendations, Assessment, Development and Evaluations (GRADE) approach. Following the confirmation of IgE-mediated food allergy diagnosis, allergen avoidance and dietary advice (with support of a specialised dietitian, if possible) together with the provision of a written treatment plan, education on the recognition of allergic symptoms and prescription of medication including adrenaline using an auto-injector are essential.
Abstract
Idiopathic hypereosinophilic syndrome (IHES) is defined by peripheral eosinophilia of more than 1500 eosinophils/µL for a 6-month duration with signs of organ involvement in the absence of parasitic infection, allergic reaction, or other recognized causes of eosinophilia.
 |
| Portal inflammation with eosinophilic infiltration. |
Key Points
Question What is the efficacy and safety of once-daily roflumilast cream, 0.15%, in patients with atopic dermatitis (AD)?
Findings In 2 phase 3 randomized clinical trials of 1337 individuals with AD, significantly more patients treated with once-daily roflumilast cream, 0.15%, achieved Validated Investigator Global Assessment for Atopic Dermatitis success at 4 weeks than patients treated with vehicle cream. Roflumilast was well tolerated with low rates of adverse events in both trials.
Meaning An effective, well-tolerated, once-daily, nonsteroidal treatment like roflumilast cream, 0.15%, may address several unmet needs and substantially improve the treatment landscape for patients with AD.
Abstract
Importance Safe, effective, and well-tolerated topical treatment options available for long-term use in patients with atopic dermatitis (AD) are limited and associated with low adherence rates.

https://doi.org/10.2147/JAA.S477376
Abstract:
Background: Adenoid hypertrophy (AH) and allergic rhinitis (AR) are common pediatric diseases, seriously affecting the quality of life and growth of children. The recurrence rate of AH is higher for patients with than for those without concurrent AR. Allergen specific immunotherapy (AIT) is the only effective therapy for modifying the course of allergic diseases. This study sought to investigate the efficacy of AIT in preventing AH recurrence in patients with AR who underwent adenoidectomy.
Methods: This study included 134 children aged 5– 12 years with concurrent AH and AR. They were separated into the subcutaneous immunotherapy (SCIT) group treated with a double-mite allergen preparation or the non-AIT group treated symptomatically with only medications. The adenoid/nasopharyngeal ratio at one year after adenoidectomy was used to assess AH recurrence.
Holze, J., Lauber, F., Soler, S. et al. Nat Commun 15, 9554 (2024). https://doi.org/10.1038/s41467-024-53770-9
Abstract
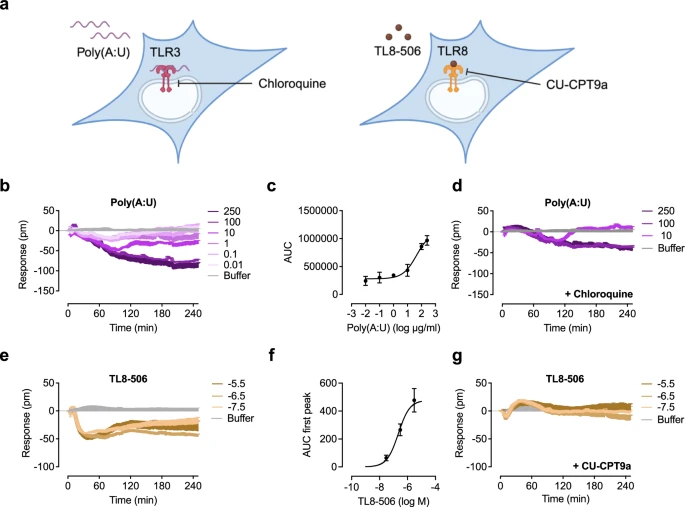 |
| Optical biosensor decodes signaling of endosomal TLRs. |
Frias Sartorelli de Toledo Piza C, Aranda CS, Solé D, Jolles S, Condino-Neto A. Front Immunol. 2024 Oct 23;15:1495564. doi: 10.3389/fimmu.2024.1495564.
Abstract
Purpose: Calculated globulin (CG, total protein minus albumin levels) correlate well with IgG levels and has been proposed as a suitable screening method for individuals with primary antibody deficiencies (PADs). We aimed to show the correlation of CG with IgG levels in children and adolescents, utilizing a common method for albumin measurement, bromocresol green.
Methods: Individuals from two Allergy and Immunology clinics were invited to participate. Inclusion criteria were age < 18, stable conditions, and signed informed consent. We included 1084 individuals. Immunoglobulin G values were determined by immunoturbidimetry; the colorimetric bromocresol green method and the Architect Biuret method were utilized for the albumin and total protein (TP) measurements, respectively.
Abstract
Dermatophagoides pteronyssinus (Der p) subcutaneous immunotherapy (SCIT) has demonstrated efficacy in clinical trials of childhood allergic rhinitis (AR). Currently, there is a lack of some generally accepted biomarkers that may predict the clinical response to SCIT to eventually achieve personalized therapy. In this study, 28 children with AR received Der p SCIT for 26–30 months at baseline, and four efficacy endpoints, serum interleukin (IL)-5, periostin, Der p-specific IgE (sIgE), and Der p sIgG4, were measured by ELSIA. Clinical symptoms and characteristics were assessed by questionnaires, and the associations among periostin, Der p 2 sIgE and clinical efficacy were analyzed. The results showed that SCIT demonstrated a significant reduction in Der p 1 sIgE (P < 0.05) and Der p 2 sIgE (P < 0.01), an increase in Der p sIgG4 (P < 0.001) and an improvement in clinical efficacy at the fourth efficacy endpoint compared with that at baseline.
 |
| Correlations between the changes in clinical indices in childhood with AR treatment with SCIT. |