Yong, M., Kirubalingam, K., Desrosiers, M.Y. et al. Allergy Asthma Clin Immunol 19, 90 (2023). https://doi.org/10.1186/s13223-023-00823-1
Abstract
Background
Dupilumab, omalizumab, and mepolizumab are the three biologics currently approved for use in CRSwNP in Canada. Despite evidence of efficacy, their cost-effectiveness, which is a key factor influencing prescribing patterns, has not yet been compared to each other.
Methods
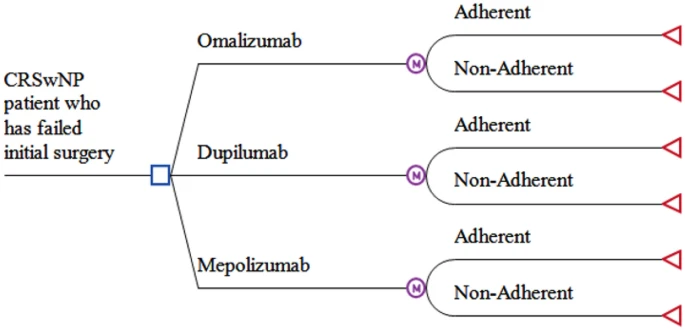
A cost-effectiveness model using quality-adjusted life years (QALYs) was constructed using a Decision Tree Markov analysis. A third-party healthcare payer perspective and a 10-year time horizon was used. A willingness-to-pay (WTP) threshold of 50,000 Canadian dollars (CAD) per QALY was used to determine cost-effectiveness. Dupilumab, omalizumab, and mepolizumab were each compared to each other.
Results
Omalizumab was the most cost-effective biologic using current estimates of cost and efficacy in CRSwNP. Using omalizumab as a baseline, dupilumab had an ICER of $235,305/QALY. Mepolizumab was dominated by omalizumab and dupilumab at the current drug prices and estimates of efficacy. Sensitivity analyses determined that when increasing the WTP threshold to $150,000/QALY, dupilumab became cost-effective compared to omalizumab in 22.5% of simulation scenarios. Additionally, altering dosing frequency had a significant effect on cost-effectiveness.
Conclusion
When comparing the relative cost-effectiveness of biologics in recalcitrant CRSwNP, omalizumab currently appears to be the most cost-effective option. Future reductions in drug prices, adjustments to currently approved dosing regimens, better patient selection, and improvements in sinus surgery outcomes will challenge the current cost-effectiveness models and necessitate reassessment as treatments for CRSwNP continue to evolve.

No comments:
Post a Comment