Hiroyuki Yagyu, Masahiro Kiuchi, Kiyoshi Hirahara. International Immunology, 2026;, dxag015, https://doi.org/10.1093/intimm/dxag015
Abstract
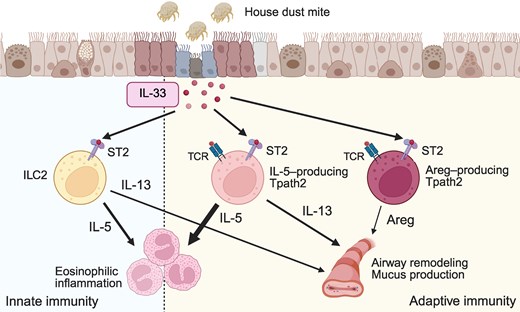 |
| IL-33-mediated activation of innate and adaptive type 2 immune cells. |
Immune memory is central to host protection against pathogens and contributes to the pathogenesis of chronic inflammatory diseases. Beyond canonical cytokines and antigenic stimuli, metabolic signals have emerged as pivotal regulators of T-cell activation, differentiation, and memory formation. However, the mechanisms by which metabolic and tissue-derived cues imprint pathogenic features on T cells remain poorly understood. Dysregulation of type 2 immune memory is a major driver of chronic inflammation in allergic diseases. A distinct subpopulation of memory T helper 2 (Th2) cells that express the interleukin (IL)-33 receptor ST2 has emerged as a key contributor to the pathogenesis of chronic allergic inflammation. Recent studies have revealed a close link between lipid metabolism and pathogenic type 2 immunity, highlighting how fatty acid uptake, storage within lipid droplets, and catabolism through lipolysis and lipophagy regulate the expression of ST2 and cytokine production. Moreover, transcriptional regulators such as peroxisome proliferator-activated receptor γ (PPARγ) integrate lipid-derived signals with transcriptional and metabolic programs that induce pathogenic memory. In this review, we summarize the current understanding of CD4+ T cell-mediated immune memory, with a particular focus on pathogenic Th2 cells and airway type 2 immunity, and discuss emerging concepts that connect lipid metabolic programs, transcriptional regulation, and chronic allergic inflammation.PDF


No comments:
Post a Comment