Sánchez, J., Caraballo, A. & Álvarez, L. Curr Treat Options Allergy 13, 3 (2026). https://doi.org/10.1007/s40521-026-00411-9
Abstract
Purpose of Review
To summarize current evidence on the immediate and long-term safety profile of the available monoclonal biological therapy (MBT) approved for the treatment of severe asthma.
Recent Findings
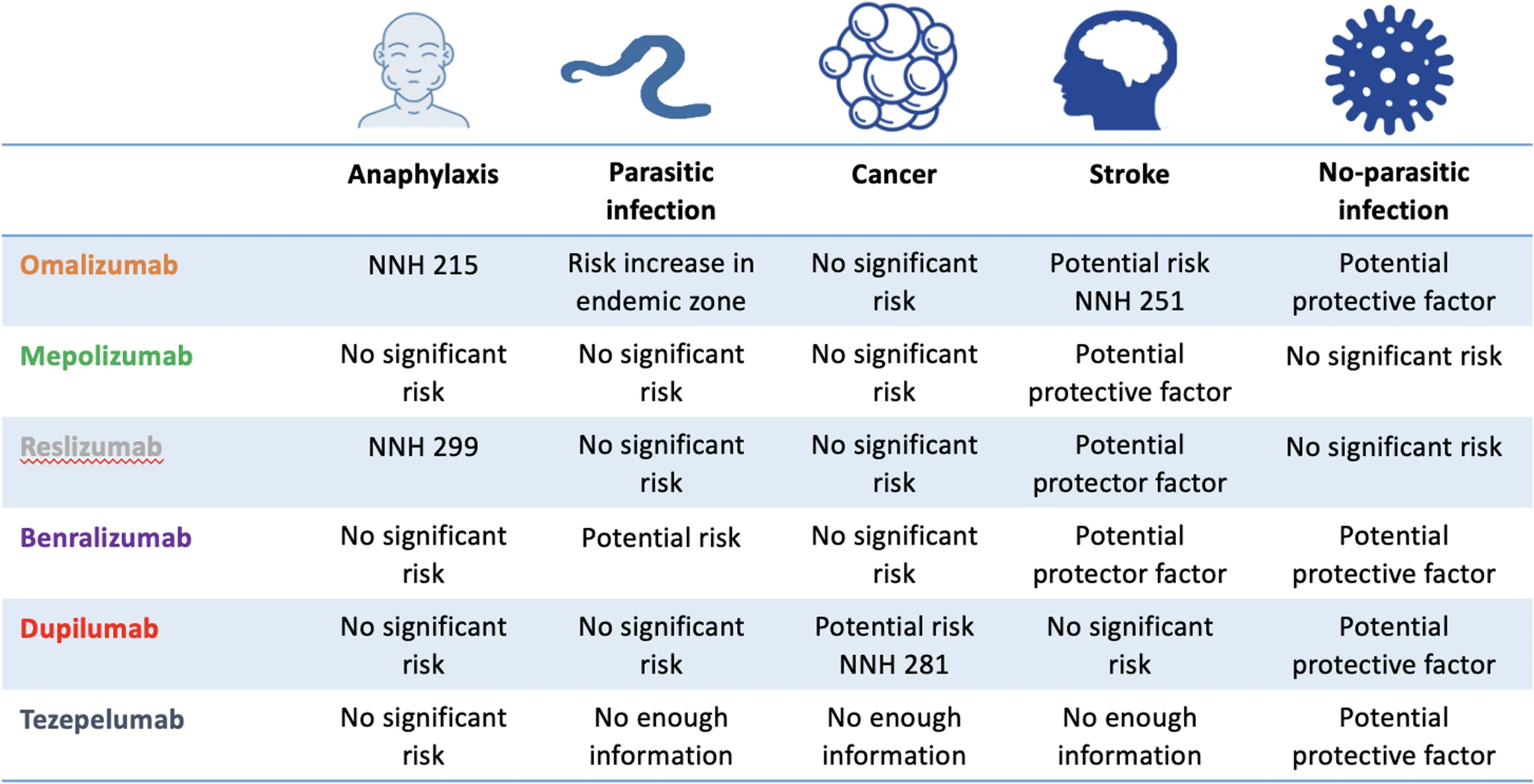 |
| Risk of serious adverse events |
Summary
MBT have significantly improved the management of severe asthma, yet continuous monitoring of their safety profile remains essential to optimize long-term outcomes and minimize risks.

No comments:
Post a Comment