 |
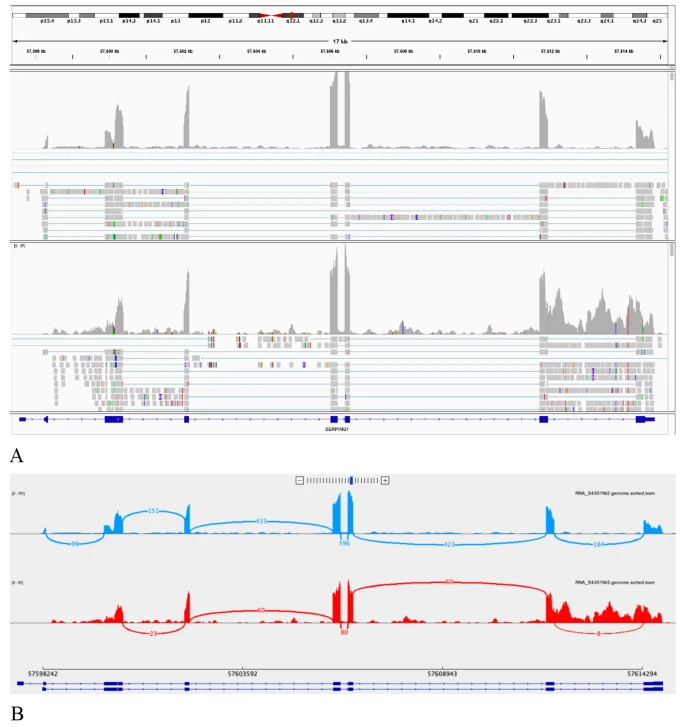
| Retention of the last SERPING1 intron present due to the NM_000062.2:c.1249 + 4 A > G r.spl variant found in patient no. 9 using transcriptome sequencing. |
A blog that publishes updates and open access scientific papers about allergy, asthma and immunology. Editor: Juan Carlos Ivancevich, MD. Specialist in Allergy & Immunology
April 2, 2024
Hereditary or acquired? Comprehensive genetic testing assists in stratifying angioedema patients
March 31, 2024
A novel quinoline with airway relaxant effects and anti-inflammatory properties
Research - Open access
Bergwik, J., Liu, J., Padra, M. et al. Respir Res 25, 146 (2024). https://doi.org/10.1186/s12931-024-02780-8
Abstract
Background
In chronic pulmonary diseases characterized by inflammation and airway obstruction, such as asthma and COPD, there are unmet needs for improved treatment. Quinolines is a group of small heterocyclic compounds that have a broad range of pharmacological properties. Here, we investigated the airway relaxant and anti-inflammatory properties of a novel quinoline (RCD405).
Methods
The airway relaxant effect of RCD405 was examined in isolated airways from humans, dogs, rats and mice. Murine models of ovalbumin (OVA)-induced allergic asthma and LPS-induced airway inflammation were used to study the effects in vivo. RCD405 (10 mg/kg) or, for comparisons in selected studies, budesonide (3 mg/kg), were administered intratracheally 1 h prior to each challenge. Airway responsiveness was determined using methacholine provocation. Immune cell recruitment to bronchi was measured using flow cytometry and histological analyses were applied to investigate cell influx and goblet cell hyperplasia of the airways. Furthermore, production of cytokines and chemokines was measured using a multiplex immunoassay. The expression levels of asthma-related genes in murine lung tissue were determined by PCR. The involvement of NF-κB and metabolic activity was measured in the human monocytic cell line THP-1.
Results
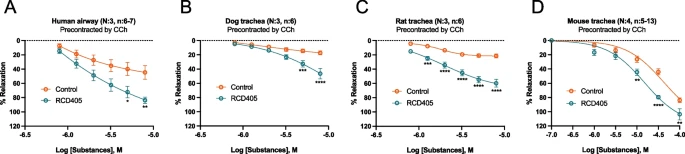 |
| The relaxing effect of RCD405 on human, dog, rat and mouse isolated airway pre-contracted with CCh. |
March 30, 2024
Clinical Practice Guideline: Immunotherapy for Inhalant Allergy
Abstract
Objective
Allergen immunotherapy (AIT) is the therapeutic exposure to an allergen or allergens selected by clinical assessment and allergy testing to decrease allergic symptoms and induce immunologic tolerance. Inhalant AIT is administered to millions of patients for allergic rhinitis (AR) and allergic asthma (AA) and is most commonly delivered as subcutaneous immunotherapy (SCIT) or sublingual immunotherapy (SLIT). Despite its widespread use, there is variability in the initiation and delivery of safe and effective immunotherapy, and there are opportunities for evidence-based recommendations for improved patient care.
Purpose
The purpose of this clinical practice guideline (CPG) is to identify quality improvement opportunities and provide clinicians trustworthy, evidence-based recommendations regarding the management of inhaled allergies with immunotherapy. Specific goals of the guideline are to optimize patient care, promote safe and effective therapy, reduce unjustified variations in care, and reduce the risk of harm. The target patients for the guideline are any individuals aged 5 years and older with AR, with or without AA, who are either candidates for immunotherapy or treated with immunotherapy for their inhalant allergies. The target audience is all clinicians involved in the administration of immunotherapy.
March 27, 2024
Real-world association between systemic corticosteroid exposure and complications in US patients with severe asthma
Research - Open access
Thomas B Casale, Thomas Corbridge, Guillaume Germain, François Laliberté, Sean D MacKnight, Julien Boudreau, Mei S Duh & Arijita Deb. Allergy, Asthma & Clinical Immunology volume 20, Article number: 25 (2024)
Abstract
Background
Systemic corticosteroid (SCS) use remains widespread among patients with severe asthma, despite associated complications.
Objective
Evaluate the association between cumulative SCS exposure and SCS-related complications in severe asthma.
Methods
This retrospective, longitudinal study used claims data from the Optum Clinformatics Data Mart database (GSK ID: 214469). Eligible patients (≥ 12 years old) had an asthma diagnosis and were divided into two cohorts: SCS use and non/burst-SCS use. Patients in the SCS use cohort had a claim for a daily prednisone-equivalent dose ≥ 5 mg SCS following ≥ 6 months of continuous SCS use; those in the non/burst-SCS cohort had no evidence of continuous SCS use and had a non-SCS controller/rescue medication initiation claim. For each cohort, the date of the qualifying claim was the index date. SCS users were further stratified by SCS use during each quarter of follow-up: low (≤ 6 mg/day), medium (> 6–12 mg/day), high (> 12 mg/day), and continuous high (≥ 20 mg/day for 90 days).
March 26, 2024
IgE to cyclophilins in pollen allergic children: epidemiological, clinical and diagnostic relevance of a neglected panallergen
Matricardi PM, Potapova E, Panetta V et al. J Allergy Clin Immunol. 2024 Mar 19:S0091-6749(24)00235-5. doi: 10.1016/j.jaci.2024.01.030.
Abstract
Background: - Cyclophilins are ubiquitous panallergens whose epidemiological, diagnostic, and clinical relevance is largely unknown and whose sensitization is rarely examined in routine allergy practice. The aim of this study was to investigate the epidemiological, diagnostic, and clinical relevance of cyclophilin in seasonal allergic rhinitis and its comorbidities.
Methods: - We examined a random sample (25%, n 253) of 1263 Italian children affected by seasonal allergic rhinitis from the "Panallergen in Pediatrics" (PAN-PED) cohort. Patients' disease phenotype had been already fully characterized through questionnaires (ARIA), skin prick tests (ALK), IgE tests to extracts, major and cross-reactive allergenic molecules of a comprehensive variety of allergenic pollen (immunoCAP), and carbohydrate cross-reacting determinants (CCD) (NOVEOS).
 |
| Graphical Abstract |
March 25, 2024
The role of molecular diagnosis in anaphylactic patients with dual or triple-sensitization to Hymenoptera venoms
Bemanian, M.H., Shokouhi Shoormasti, R., Arshi, S. et al. Allergy Asthma Clin Immunol 20, 22 (2024). https://doi.org/10.1186/s13223-024-00885-9
Abstract
Background
The poly-sensitization to Hymenoptera venom makes it difficult to select genuine allergens for immunotherapy and increases patients’ costs. The objective of this study was to determine the culprit allergen in dual or triple-sensitized patients to three Hymenoptera venoms through molecular diagnosis and evaluating the results of incorporating the molecular diagnosis with skin tests.
Methods
Thirty-two patients with anaphylactic reactions and dual or triple-sensitization to Hymenoptera venoms in skin tests entered this study. IgE-sensitization to whole extracts and molecules of Apis mellifera (Api m), Vespula vulgaris (Ves v), and Polistes dominulus (Pol d) was evaluated utilizing ALEX or ImmunoCAP.
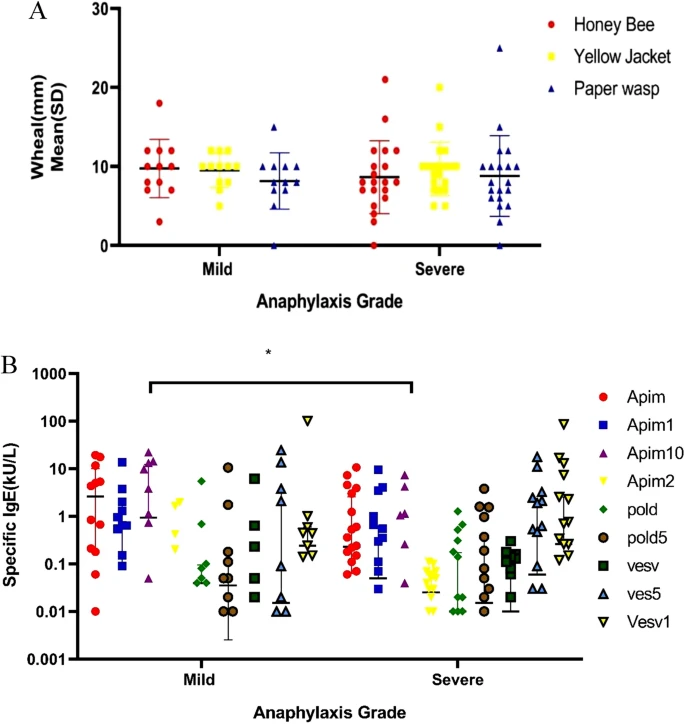
Results
Twenty-nine patients (90.6%) were male. IgE-sensitization to at least one of the allergenic molecules related to Apis mellifera, Vespula vulgaris, and Polistes dominulus was seen in 59.4, 53.1, and 21.9%, respectively. Among 32 patients, 14 (43.8) and 8 (25%), were mono-sensitized to Api m and Ves v components in ALEX, respectively. Double sensitization to Hymenoptera was identified in 18.8% of patients in ALEX.Navigating Food Allergies: Advances in Diagnosis and Treatment Strategies
Abstract
Food allergy is a major health concern worldwide, encompassing both immunologic and non-immunologic reactions. This review thoroughly examines the pathophysiology, clinical manifestations, and treatment options for various types of food allergies. Immunologic food allergies, including IgE-mediated reactions such as oral allergy syndrome and systemic anaphylaxis, pose various diagnostic and management challenges. Non-IgE-mediated reactions such as food protein-induced enterocolitis syndrome, dermatitis herpetiformis, and proctocolitis necessitate individualized patient care. In addition, mixed reactions such as eosinophilic esophagitis and atopic dermatitis complicate the clinical picture.
March 24, 2024
Lupin, a potential "hidden" food anaphylaxis allergen: An alert from the Allergy-Vigilance Network®.
To the Editor,
Lupin is a legume considered as an emerging food allergen, but the true prevalence of lupin allergy is not known.1 Due to the increasing consumption of lupin and its potential cross-reactivity with peanut, the Allergy-Vigilance Network® (AVN) aimed to report lupin-induced anaphylaxis cases and compare their main characteristics to other foods (See Data S1 for Material and Methods).
 |
| Main characteristics of 62 patients with a lupin-induced anaphylaxis recorded by the Allergy-Vigilance Network® and comparisons by age groups |