Kim D, Cho HJ, Kim CH, Rha MS. Allergol Int. 2025 Oct 15:S1323-8930(25)00109-1. doi: 10.1016/j.alit.2025.09.003.
Abstract
Background
Methods
Results
 |
| Top allergen loadings for each non-negative matrix factorization signature. |
A blog that publishes updates and open access scientific papers about allergy, asthma and immunology. Editor: Juan Carlos Ivancevich, MD. Specialist in Allergy & Immunology
Kim D, Cho HJ, Kim CH, Rha MS. Allergol Int. 2025 Oct 15:S1323-8930(25)00109-1. doi: 10.1016/j.alit.2025.09.003.
 |
| Top allergen loadings for each non-negative matrix factorization signature. |
Abstract
Importance Artificial intelligence (AI) is changing health and health care on an unprecedented scale. Though the potential benefits are massive, so are the risks. The JAMA Summit on AI discussed how health and health care AI should be developed, evaluated, regulated, disseminated, and monitored.
Observations Health and health care AI is wide-ranging, including clinical tools (eg, sepsis alerts or diabetic retinopathy screening software), technologies used by individuals with health concerns (eg, mobile health apps), tools used by health care systems to improve business operations (eg, revenue cycle management or scheduling), and hybrid tools supporting both business operations (eg, documentation and billing) and clinical activities (eg, suggesting diagnoses or treatment plans). Many AI tools are already widely adopted, especially for medical imaging, mobile health, health care business operations, and hybrid functions like scribing outpatient visits. All these tools can have important health effects (good or bad), but these effects are often not quantified because evaluations are extremely challenging or not required, in part because many are outside the US Food and Drug Administration’s regulatory oversight. A major challenge in evaluation is that a tool’s effects are highly dependent on the human-computer interface, user training, and setting in which the tool is used.
He Y, Liu X, Zhou B, Dai T. Front Immunol. 2025 Sep 25;16:1658826. doi: 10.3389/fimmu.2025.1658826.
Objective: The aim of this study was to summarize the therapeutic efficacy of various allergen immunotherapy (AIT) in the treatment of allergic rhinitis (AR) among different populations and for different allergens.
Methods: Systematic reviews or meta-analyses related to the efficacy of AIT in the treatment of AR until October 2024 were retrieved from PubMed, Web of Science, Embase, and Cochrane Library. Each study was independently evaluated by two investigators in accordance with the inclusion and exclusion criteria. The methodological quality was assessed using AMSTAR 2, and the quality of evidence was evaluated by the GRADE system.
 |
| Forest plot of the efficacy of SLIT in AR patients allergic to HDM. [(A) Symptom scores. (B) Medication scores]. |
Abstract
Introduction: Uncontrolled asthma greatly affects quality of life globally and highlights unmet medical needs. Despite advances in treatment and care, many patients still experience delayed diagnoses, poor symptom control, and a reliance on emergency care. The Global Allergy and Airways Patient Platform (GAAPP) surveyed patients with moderate-to-severe uncontrolled asthma to assess their care experiences.
Methods: The GAAPP Time Clock Survey is a cross-sectional, online, multilingual survey of adults living in Brazil, Germany, Italy, Japan, Saudi Arabia, the United Arab Emirates, and the US. The survey examined diagnosis, symptoms, treatment outcomes, challenges in self-management, and timelines for care coordination.
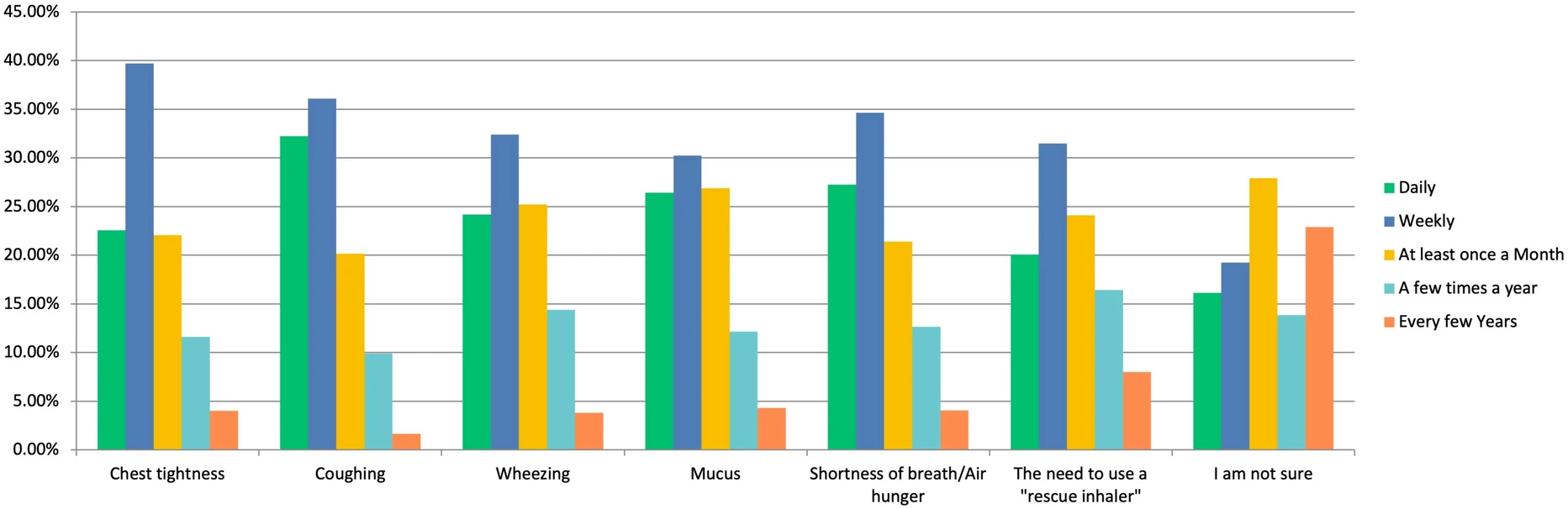 |
| Respondents’ Symptom Report Self-reported frequency of common asthma symptoms, including cough, shortness of breath, mucus production, wheezing, chest tightness, and rescue inhaler use |
Feleszko W, Caminati M, Gern JE et al. Ann Allergy Asthma Immunol. 2025 Oct 8:S1081-1206(25)01201-3. doi: 10.1016/j.anai.2025.09.015.
Abstract
Background
Tezepelumab, a human monoclonal antibody, blocks the activity of thymic stromal lymphopoietin (TSLP). In the phase 2b PATHWAY (NCT02054130) and phase 3 NAVIGATOR (NCT03347279) studies, tezepelumab reduced exacerbations and improved lung function, asthma control, and health-related quality of life versus placebo in patients with severe, uncontrolled asthma.
Objective
This post hoc analysis of PATHWAY and NAVIGATOR evaluated the incidence of asthma exacerbations co-occurring with documented acute respiratory illnesses attributed to infections.
Methods
Patients were randomized 1:1 to receive tezepelumab 210 mg subcutaneously or placebo every 4 weeks for 52 weeks. The incidence of asthma exacerbations co-occurring with respiratory illness-related adverse events (AEs) was assessed. Co-occurrence was defined as at least 1 day of overlap between a respiratory illness-related AE and the asthma exacerbation period beginning 7 days before the start of the exacerbation until the end of the asthma exacerbation.
Results
 |
| Respiratory illness-related AEs occurred at an incidence of ≥ 1 per 100 patient-years in either treatment group |
Sousa-Pinto B, Savouré M, Vieira RJ et al. J Investig Allergol Clin Immunol. 2025 Oct 15;35(5):373-383. doi: 10.18176/jiaci.1047.
Abstract:
Background: The Allergic Rhinitis and its Impact on Asthma (ARIA) guidelines classify rhinitis as "intermittent" or "persistent" and "mild" or "moderate-severe".
Objectives: To assess ARIA classes in a real-world study in terms of phenotypic differences and their association with asthma.
Methods: We performed a cross-sectional real-world study based on users of the MASK-air® app who reported data for at least 3 different months. We assessed the frequency of users according to the ARIA classes and compared these classes in terms of rhinitis symptoms, use of comedication, frequency of comorbid asthma, and the association between comorbid asthma and rhinitis control.
Di Bona D, Paoletti G, Cognet-Sicé J et al. J Investig Allergol Clin Immunol 2025; Vol 35(5) : 341-352 doi: 10.18176/jiaci.1076
Abstract:
 |
| Subgroup analyses of symptom score by age and sensitization status of sublingual immunotherapy vs placebo. |
Beiner, L., Boehm, I. Insights Imaging 16, 216 (2025). https://doi.org/10.1186/s13244-025-02099-y
Abstract
 |
| Graphical Abstract |