ABSTRACT
 |
Characteristics of immediate drug hypersensitivity reactions (DHR) caused by excipients. |
A blog that publishes updates and open access scientific papers about allergy, asthma and immunology. Editor: Juan Carlos Ivancevich, MD. Specialist in Allergy & Immunology
ABSTRACT
 |
Characteristics of immediate drug hypersensitivity reactions (DHR) caused by excipients. |
Key Points
Question What are the risk factors associated with the development of food allergy in children?
Findings This systematic review and meta-analysis of 2.8 million participants in 190 studies identified the following largest and most certain risk factors associated with the development of food allergies in children: prior allergic conditions (atopic march/diathesis), atopic dermatitis, increased skin transepidermal water loss, filaggrin gene sequence variations, delayed solid food introduction, infant and intrapartum antibiotic exposure, male sex, being first born, family history of allergy, parental migration, self-identification as Black, and cesarean delivery.
Meaning This systematic review and meta-analysis clarifies the major and minor risk factors associated with developing early-onset food allergy to inform optimal prevention clinical practice, policy, and research.
Abstract
Importance The incidence and risk (predictive) factors for early life food allergy development remain uncertain.
Abstract
Purpose of Review
This review examines whether allergen immunotherapy (AIT) for grass pollen allergy should expand beyond the recent trend towards a mono-species approach based on Phleum pratense. It explores whether multi-species formulations better reflect natural exposure and could improve clinical outcomes.
Recent Findings
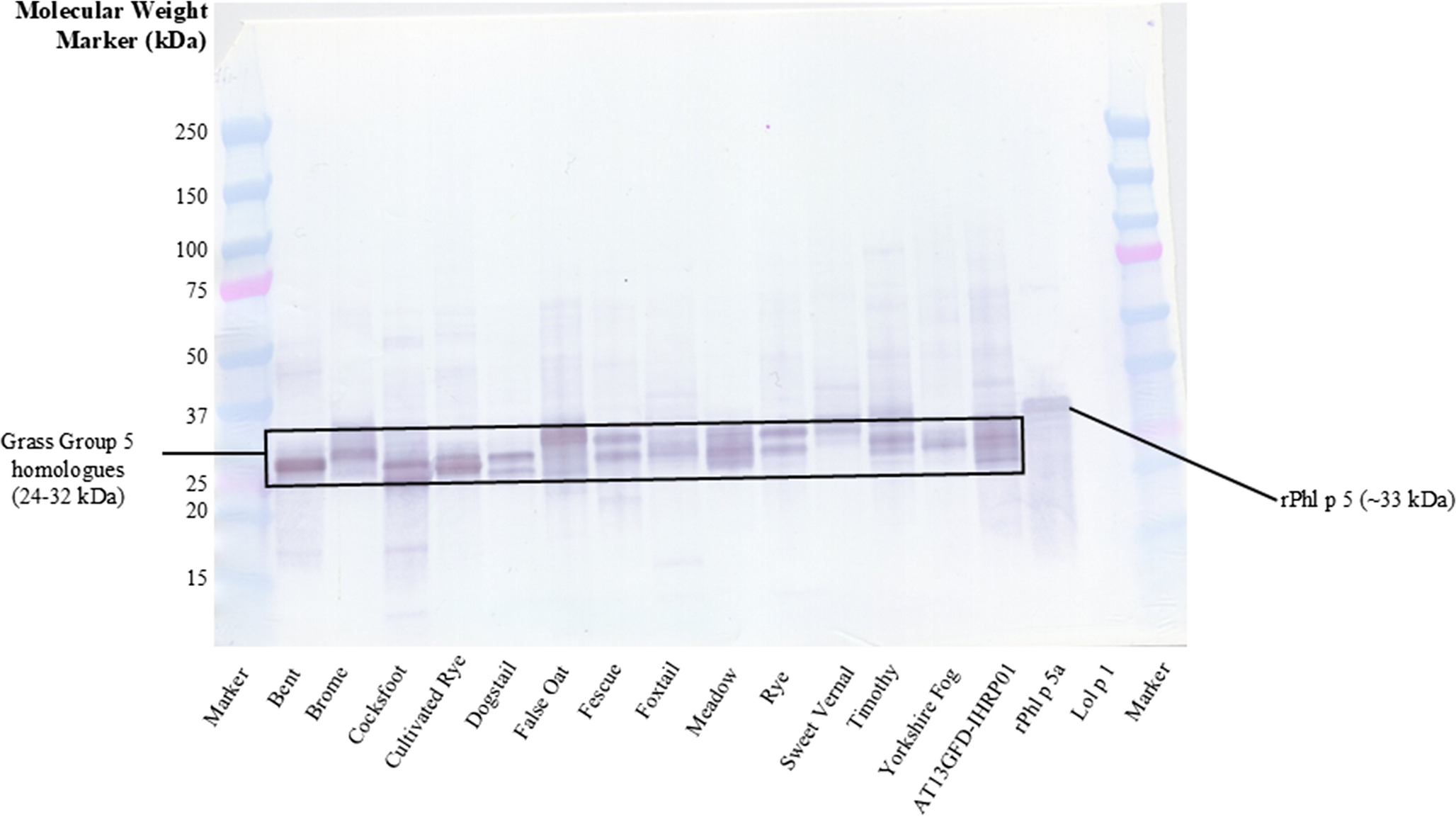 |
| Group 5 homologues identified in individual extracts and a mixed extract of 13 species of Poaceae family grasses, using a monoclonal antibody |
Sánchez, J., Caraballo, A. & Álvarez, L. Curr Treat Options Allergy 13, 3 (2026). https://doi.org/10.1007/s40521-026-00411-9
Abstract
Purpose of Review
To summarize current evidence on the immediate and long-term safety profile of the available monoclonal biological therapy (MBT) approved for the treatment of severe asthma.
Recent Findings
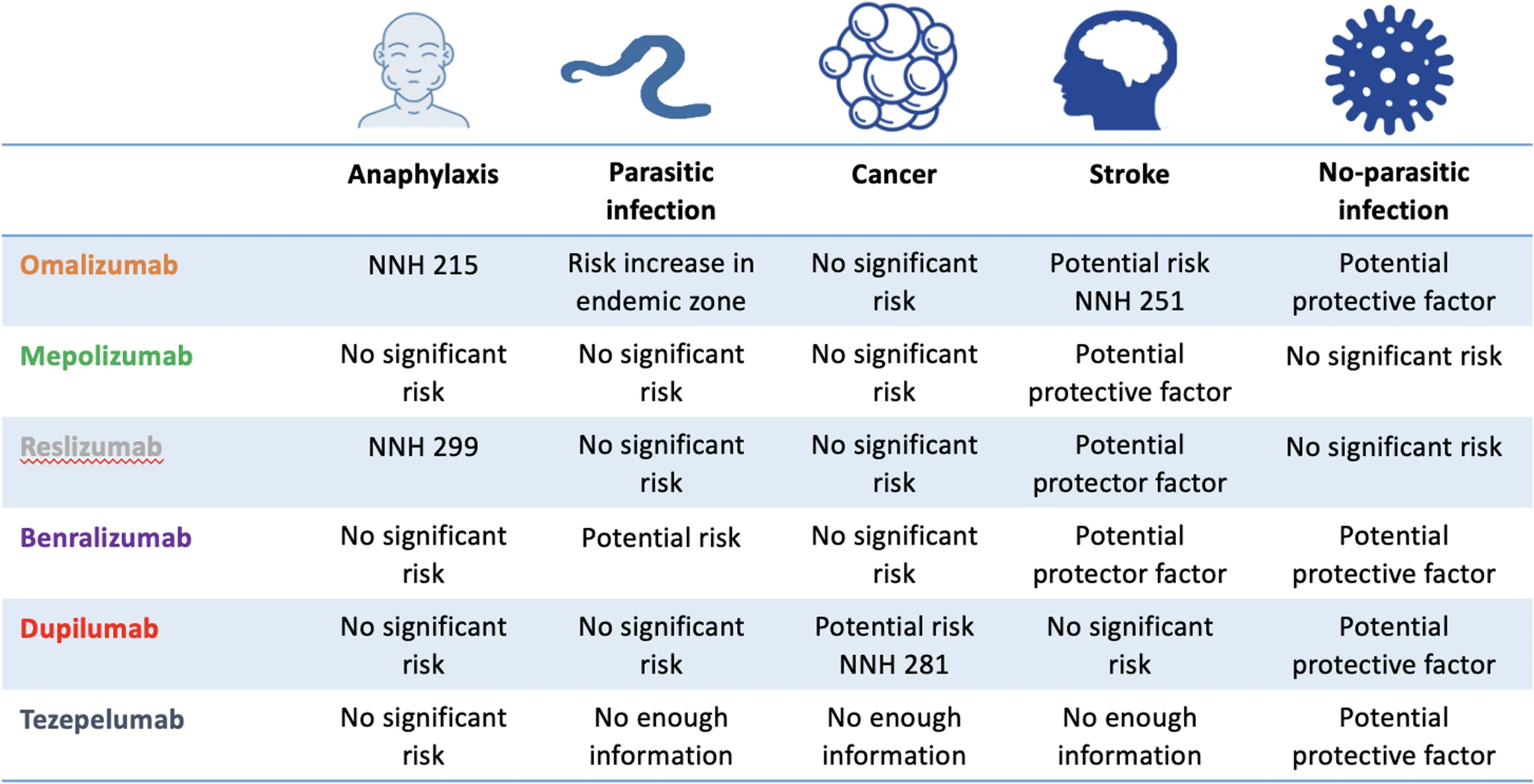 |
| Risk of serious adverse events |
Abstract
Background
Enecilla MLB, Recto MST, Navarro-Locsin CG et al. Asia Pac Allergy. 2026 Feb;16(1):20-29. doi: 10.5415/apallergy.0000000000000214.
Abstract
Background
Pediatric atopic dermatitis (AD) is a common, chronic inflammatory skin disorder that significantly impacts the quality of life of affected children and their families. Multiple therapies were approved to treat AD in children and adolescents since publication of the AAD's 2014 AD guidelines.
Objective
To provide evidence-based recommendations on the use of topical therapies, phototherapy, and systemic therapies for AD in children and adolescents.
To investigate age-related differences in efficacy and safety of subcutaneous immunotherapy (SCIT) among patients with allergic rhinitis (AR).
Retrospective cohort study.
Tertiary referral center.
AR patients who completed a 3-year course of dust mite SCIT with a 2-year post-SCIT follow-up were categorized into pediatric and adult groups. Baseline characteristics, SCIT efficacy, and adverse reactions were compared between groups. Multivariable logistic regression was used to identify independent predictors of SCIT efficacy and adverse reaction.
 |
| Comparison of SCIT efficacy between children and adults. |