Abstract
Purpose of Review
This review examines whether allergen immunotherapy (AIT) for grass pollen allergy should expand beyond the recent trend towards a mono-species approach based on Phleum pratense. It explores whether multi-species formulations better reflect natural exposure and could improve clinical outcomes.
Recent Findings
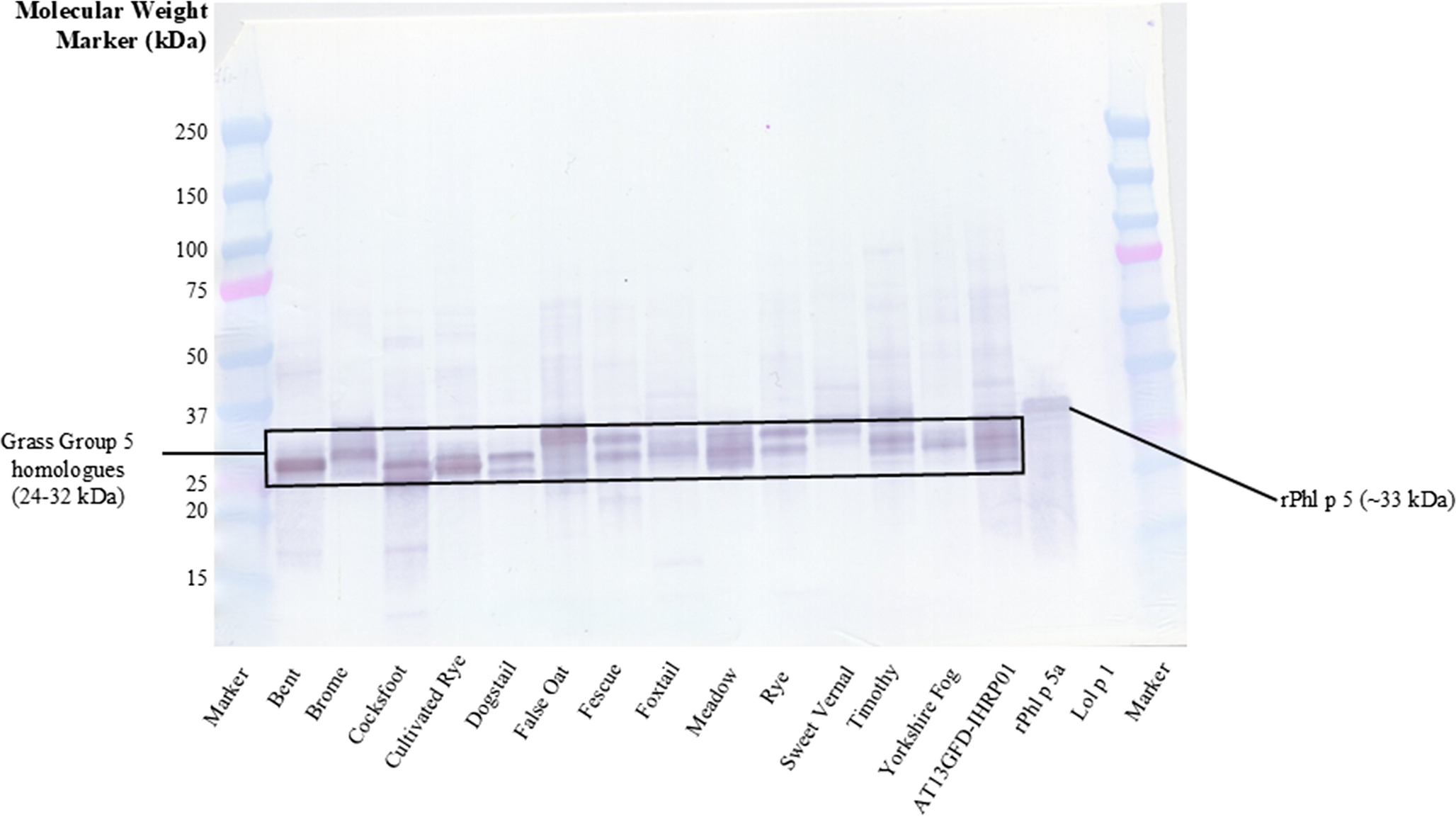 |
| Group 5 homologues identified in individual extracts and a mixed extract of 13 species of Poaceae family grasses, using a monoclonal antibody |
Research from aerobiology and immunology shows that grass pollen exposure involves diverse species with distinct flowering periods, influenced by climate and geography. Molecular analyses reveal species-specific allergen profiles, including unique peptides and variations in major allergens such as Group 1 and 5. Patient data confirm symptom variability across the season. In-vitro studies have observed limits to the cross-reactivity of T-cell epitopes, and comparative clinical studies suggest benefits for multi-species treatment options.
Summary
Evidence indicates that mono-species extracts alone do not represent the full allergenic spectrum of grass pollen. Broad-spectrum AIT formulations incorporating multiple grass species provide a more comprehensive repertoire of allergens and epitopes, potentially enhancing immunogenicity and therapeutic benefit. This supports the hypothesis that diversity does not equate to dilution in broad-spectrum formulations. The approach aligns with patient symptom patterns and may improve efficacy and asthma prevention. Future research could further refine species selection and leverage molecular diversity to optimize treatment strategies.

No comments:
Post a Comment