Wang X, Zhou N, Zhi Y. J Asthma Allergy. 2025;18:539-544
https://doi.org/10.2147/JAA.S509939
 |
| (A) and (B) Urticaria-like rash on legs and right arm |
A blog that publishes updates and open access scientific papers about allergy, asthma and immunology. Editor: Juan Carlos Ivancevich, MD. Specialist in Allergy & Immunology
Wang X, Zhou N, Zhi Y. J Asthma Allergy. 2025;18:539-544
https://doi.org/10.2147/JAA.S509939
 |
| (A) and (B) Urticaria-like rash on legs and right arm |
Abstract
Allergic diseases, including allergic conjunctivitis (AC), pose a significant health burden, affecting both developed and developing nations. Despite its importance, AC is often underreported, leading to underestimated incidence and prevalence. The coexistence of AC with allergic rhinitis and its comorbidity with asthma underscore its clinical relevance. The prevalence of nasal symptoms with eye symptoms related to eye allergy varies among different age groups and regions worldwide. Climatic factors, aeroallergens, and environmental exposure play significant roles in the prevalence of ocular allergies. Allergen immunotherapy (AIT) represents the only disease-modifying treatment for IgE-mediated allergic diseases. This review provides a comprehensive overview of the history, mechanisms, and evidence of AIT for ocular allergies, with a focus on AC.
Shamriz, O., Parnasa, E., Rubin, L. et al. BMC Immunol 26, 29 (2025). https://doi.org/10.1186/s12865-025-00705-8
Abstract
Background
Physicians may encounter situations where they need to co-administer omalizumab with non–IgE-targeting monoclonal antibodies. In this study, we share our experience with these dual biologic treatments.
Objective
To evaluate the efficacy and safety of dual biological therapy using omalizumab and non-IgE-targeting monoclonal antibodies at a single center.
Abstract
Background
Limited data exist on the comparative risk of infections during biologic and Janus kinase inhibitor (JAKi) treatment for atopic dermatitis (AD) in daily practice.
Objective
To assess the differential infection risk of biologic and JAKi treatment in patients with moderate-to-severe AD in a real-world setting.
Methods
This prospective, multicentre study evaluated treatment-emergent infections in patients (age ≥ 12 years) using biologics or JAKi from the BioDay registry from October 2017 to July 2024. Crude incidence rates were calculated per 100 patient-years (PY) per treatment. Cox regression for recurrent events, adjusted for potential confounders, was used to estimate hazard ratios (HR) for the rate of infections, with subgroup and sensitivity analyses in bio-/JAKi-naïve patients.
Results
 |
| Graphical Abstract |
Abstract
Background
Severe asthma and moderate-to-severe atopic dermatitis can significantly impact the lives of children and adolescents. However, real-world data on pediatric patients’ perceptions of their medication are limited.
Methods
This non-interventional cross-sectional study at a university hospital explored patients’ perceptions. We included patients aged between 6 and 17 with severe asthma and/or moderate-to-severe atopic dermatitis. For patients treated with dupilumab, a minimum dupilumab treatment duration of 16 weeks was required. We conducted one structured interview per patient, based on a questionnaire consisting of open questions and ratings on 6-point Likert scales (response scale range: “0: not at all” to “5: very strongly”).
Results
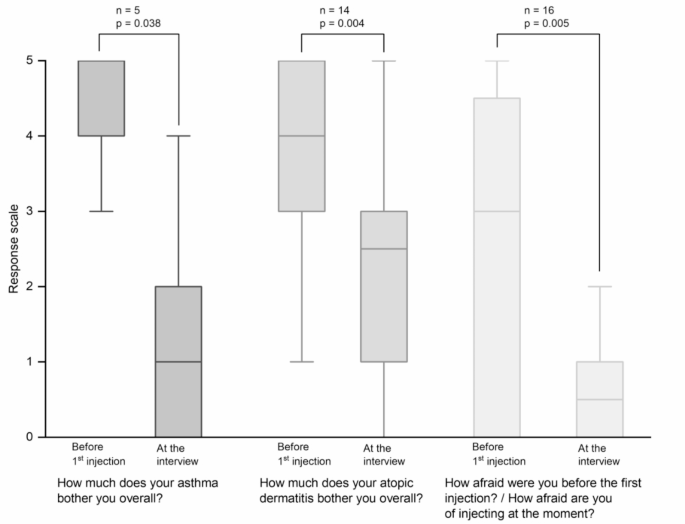 |
| Participants’ perceptions of their diseases before and during dupilumab therapy |
Zhang, Y., Li, J., Wang, M. et al. Nat Med (2025). https://doi.org/10.1038/s41591-025-03651-5
Abstract
Seasonal allergic rhinitis (SAR) places a significant socioeconomic burden, particularly on individuals with poorly managed recurrent and severe symptoms despite standard-of-care treatment. Stapokibart, a humanized monoclonal antibody that targets the interleukin (IL)-4 receptor subunit alpha, inhibits its interaction with both IL-4 and IL-13 in type 2 inflammation.
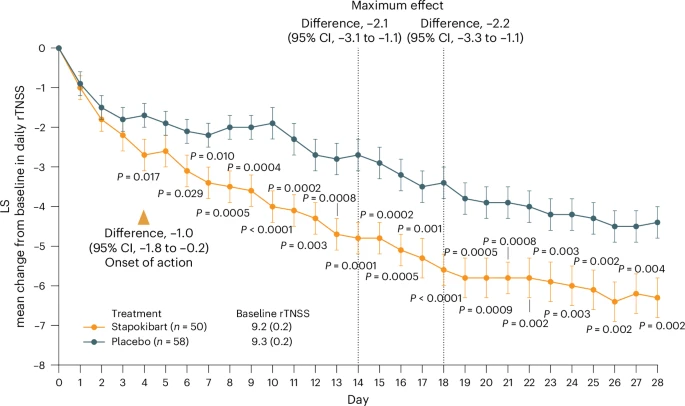 |
| Change from baseline over time in daily rTNSS during the 4-week treatment period. |
Abstract
Background
Inpatient polysomnography (PSG) is the gold standard for the diagnosis of obstructive sleep apnoea (OSA), however, both complexity and costs limit the availability of this examination. Home sleep apnoea testing devices are a diagnostic alternative in patients with increased risk of OSA. We evaluated the diagnostic performance of a Doppler radar technology based, contactless sleep apnoea testing device (CSATD) in a cohort of patients with a clinically increased risk of OSA.
Methods
Monocentric prospective study. Sleep monitoring with the CSATD SleepizOne + without pulse oximetry (Sleepiz AG, Switzerland) was performed simultaneously with elective inpatient PSG. PSG was analysed blinded to the CSATD results and according to AASM 2012 criteria by certified sleep physicians. The CSATD data were analysed automatically and independently by a dedicated software.
Cui, Z., Chen, X., Zhai, S. et al. Naunyn-Schmiedeberg's Arch Pharmacol (2025). https://doi.org/10.1007/s00210-025-04073-3
Abstract
Specific immunotherapy (SIT) is key in allergic diseases, tumor immunity, and autoimmune regulation. In recent years, the mechanism of action of drugs in SIT has attracted much attention, including the induction of hypersensitivity responses and modulation of immune tolerance. However, scientific challenges remain regarding their mechanism of action and optimization strategies. Studies on pharmacological SIT have been accumulated in the past, and there is an urgent need for bibliometric analyses to review and prospect these results for future academic development. Strict search criteria were developed to screen and download literature information from the Web of Science Core Collection.